7071. Pelargonidin
Nomenclature
Description and references
The aglucone of pelargonin: Willst"atter, Bolton, Ann. 408, 42 (1914). Synthesis: Malkin, Robinson, J. Chem. Soc. 127, 1190 (1925); Robertson et al., 1928, 1533. Prepn from kaempferol: Mirza, Robinson, Nature 166, 997 (1950); King, White, J. Chem. Soc. 1957, 3901.
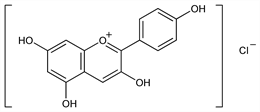
Properties
Reddish-brown prisms from 2% HCl or from alcoholic HCl. Not melted at 350°. Absorption max (ethanol + 0.01% HCl): 530 nm (ε 32,000). Sol in alcohol, methanol; moderately sol in water; slightly sol in chloroform.Derivative
3,5-Diglucoside.Nomenclature
Description and references
From flowers of Pelargonium zonale Ait. var. meteor, Geraniaceae: Willst"atter, Bolton, loc. cit.; from scarlet roses: Harborne, Experientia 17, 72 (1961). Identity with monardin and salvinin: Robinson, Todd, J. Chem. Soc. 1932, 2488. Identity with punicin: Karrer, Widmer, Helv. Chim. Acta 10, 67 (1927). Structure: Leon et al., J. Chem. Soc. 1931, 2672.
Properties
Red needles with green luster from methanol + HCl, dec 175-180°. [α]D -291°. Absorption max (methanol + HCl): 269, 505 nm. Sol in water, alc.Derivative
3-Glucoside.Nomenclature
Description and references
From purple-red aster (Callistephus chinensis (L.) Nees, Compositae): Willst"atter, Burdick, Ann. 412, 149 (1916); from strawberries: Sondheimer, Kertesz, J. Am. Chem. Soc. 70, 3476 (1948). Structure and synthesis: Robertson, Robinson, J. Chem. Soc. 1928, 1460.