7074. Pellotine
Nomenclature
CAS number: 83-14-7
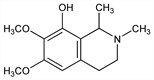
1,2,3,4-Tetrahydro-6,7-dimethoxy-1,2-dimethyl-8-isoquinolinol; N-methylanhalonidine; 8-hydroxy-6,7-dimethoxy-1,2-dimethyl-1,2,3,4-tetrahydroisoquinoline. C13H19NO3; mol wt 237.29.
C 65.80%, H 8.07%, N 5.90%, O 20.23%.
Description and references
From the mescal buttons (pellote) of Lophophora williamsii (Lemaire) Coult., Cactaceae. Structure: Sp"ath et al., Ber. 65, 1771 (1932). Synthesis: Brossi et al., Helv. Chim. Acta 47, 2089 (1964); 49, 403 (1966); Takido et al., J. Pharm. Sci. 59, 271 (1970). Biosynthetic studies: Battersby et al., Tetrahedron Lett. 1967, 563; 1968, 6111.
Properties
Plates from petr ether, mp 112°. Alkaline reaction. Freely sol in alc, acetone, ether, chloroform; sparingly sol in water.Derivative
Hydrochloride.C13H19NO3.HCl; mol wt 273.76.
C 57.04%, H 7.36%, N 5.12%, O 17.53%, Cl 12.95%.
Properties
Prisms, freely sol in water; sparingly in alcohol.Derivative
Hydriodide.C13H19NO3.HI; mol wt 365.21.
C 42.75%, H 5.52%, N 3.84%, O 13.14%, I 34.75%.