7076. β-Peltatin
Nomenclature
CAS number: 518-29-6
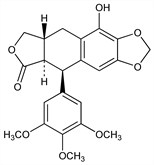
[5R-(5α,5aβ,8aα)]-5,8,8a,9-Tetrahydro-10-hydroxy-5-(3,4,5-trimethoxyphenyl)furo[3′,4′:6,7]naphtho[2,3-d]-1,3-dioxol-6(5aH)-one; 8-hydroxy-2-hydroxymethyl-6,7-methylenedioxy-4-(3′,4′,5′-trimethoxyphenyl)-1,2,3,4-tetrahydronaphthalene-3-carboxylic
acid lactone; β-peltatin A. C22H22O8; mol wt 414.41.
C 63.76%, H 5.35%, O 30.89%.
Description and references
Exists as a glucoside in the rhizomes of Podophyllum peltatum L., Berberidaceae: von Wartburg et al., Helv. Chim. Acta 40, 1331 (1957). Isoln from resin podophyllum: Hartwell, Detty, J. Am. Chem. Soc. 70, 2833 (1948); 72, 246 (1950); from Hyptis verticillata Jacq., Labiatae: German, J. Pharm. Sci. 60, 649 (1971).
Properties
Prisms from abs ethanol. Dec 238-241° (slight sintering begins at 234°). [α]D20 -122.9° (c = 0.578 in chloroform). Somewhat less sol than α-peltatin. Soly in water at 23°: 13 mg/liter. Fairly sol in chloroform, hot ethanol, acetic acid, acetone, dil caustic; less sol in benzene, ether, carbon tetrachloride, propylene glycol. Practically insol in petr ether.Derivative
β-Peltatin-β-d-glucoside.C28H32O13; mol wt 576.55.
C 58.33%, H 5.59%, O 36.08%.