Nomenclature
CAS number: 7616-94-6
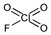
ClFO
3; mol wt 102.45.
Cl 34.61%, F 18.54%, O 46.85%.
Description and references
Reviews: Downs, Adams in Comprehensive Inorganic Chemistry vol.
2, J. C. Bailar, Jr. et al., Eds. (Pergamon Press,
Oxford, 1973) pp 1391-1393; Christe, Schack in Adv. Inorg. Chem. Radiochem. 18, 319-398.
Properties
Characteristic sweet odor. Usually stored in
cylinders as liquid under pressure. mp -147.7°. bp -46.7°. d20 (liq) 1.434. Critical temp 95.2°. Crit pressure
53 atm. Crit density: 0.637. Heat of vaporization 4.6 kcal/mol.
Trouton constant: 20.4. Dipole moment 0.03. Heat of formation
of gas at 25° -5.12 kcal/mol. Sp heat of liquid: 0.229 cal/g/°C
at -40°; 0.290 at +50°. Does not corrode base metals when anhydr.
Shows the greatest resistance to electrical breakdown known for any
gas. Handle with caution. Do not
bring in direct contact with reducing agents, alcohols, etc.: Chem. Eng. News 37, 60 (1959).
Explosions have occurred.Caution
Potential symptom of overexposure
is respiratory system irritation; direct contact with liquid may cause
frostbite. See NIOSH Pocket Guide
to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 246.
May cause methemoglobinemia, cyanosis, alveolar edema, bronchopneumonia.
See Patty's Industrial Hygiene
and Toxicology vol. 2F, G. D. Clayton, F. E.
Clayton, Eds. (John Wiley & Sons, New York, 4th ed., 1994) p 4478.Use
In organic synthesis to introduce fluorine atoms
into organic molecules. As oxidizing agent; insulator for high voltage
systems.
