7167. Perillaldehyde
Nomenclature
Description and references
Isoln from Perilla arguta Benth., Labiatae: Semmler, Zaar, Ber. 44, 52, 815 (1911); from essential oil of Sium latifolium L., Umbelliferae: Parczewski, Diss. Pharm. 12, 223 (1960), C.A. 55, 7765c (1961); from mandarin peel oil (Citrus reticulata Blanco, Rutaceae): Kugler, Kováts, Helv. Chim. Acta 46, 1480 (1963). Prepn by chromic oxidation of perilla alcohol: Naves, ibid. 29, 553 (1946); Ritter, Ginsburg, J. Am. Chem. Soc. 72, 2381 (1950); Kergomard, Philibert-Bigou, Bull. Soc. Chim. Fr. 1958, 393, 1174; Naves, Grampoloff, ibid. 1960, 37. Brief review: Food Chem. Toxicol. 20, Suppl. I-IV, 799-800 (1982).
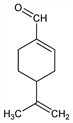
Properties
LD50 in mice (g/kg): 1.72 orally; in guinea pigs (g/kg): >5 dermally (Food Chem. Toxicol.).Derivative
d-Form.Properties
Liquid. bp745 237°; bp7 98-100°. d420 0.953. nD20 1.5058. [α]D20 +127° (c = 13.1 in carbon tetrachloride).Derivative
l-Form.Properties
Liquid. bp10 104-105°. d420 0.9645. nD20 1.5069. [α]D20 -146°.Derivative
Oxime.Nomenclature
Description and references
Previously referred to as l-perillaldehyde α-anti-oxime. Synthesis: And^o et al., Science (Tokyo) 17, 241 (1947), C.A. 45, 1976d (1951). Clarification of structure: Acton et al., Experientia 26, 473 (1970).