7176. Periplogenin
Nomenclature
CAS number: 514-39-6
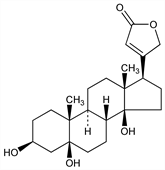
(3β,5β)-3,5,14-Trihydroxycard-20(22)-enolide; desoxostrophanthidin. C23H34O5; mol wt 390.51.
C 70.74%, H 8.78%, O 20.49%.
Description and references
The aglycon of periplocin and periplocymarin; accompanies strophanthidin in Strophanthus eminii Asch. et Pax., Apocynaceae. Isoln: Lehmann, Arch. Pharm. 235, 157 (1897); Jacobs, Hoffmann, J. Biol. Chem. 79, 519 (1928); Stoll, Renz, Helv. Chim. Acta 22, 1193 (1939); Lardon, ibid. 33, 639 (1950). Structure: Speiser, Reichstein, Experientia 3, 323 (1947); eidem, Helv. Chim. Acta 30, 2143 (1947); 31, 622 (1948). Synthesis: Deghenghi, Gaudry, Tetrahedron Lett. 1963, 2045; Kamano et al., J. Org. Chem. 39, 2319 (1974).
Properties
Solvated prisms from methanol which contain an undetermined amount of methanol, sinter at 140°, mp 235°. [α]D27 +31.5° (c = 1.04 in alcohol). Sol in alcohol and chloroform; slightly sol in ether, water (1:2500); practically insol in benzene, petr ether. pH of aq solns ≈7. Strong positive Legal test.Derivative
Monobenzoate.C30H38O6; mol wt 494.62.
C 72.85%, H 7.74%, O 19.41%.
Properties
Stout glistening wedges from 95% alcohol, mp 235°.Derivative
Dihydroperiplogenin.C23H36O5; mol wt 392.53.
C 70.38%, H 9.24%, O 20.38%.
Properties
Stout prisms from 25% alcohol, mp 204° (slight preliminary softening).Derivative
3-O-Acetylperiplogenin.C25H36O6; mol wt 432.55.
C 69.42%, H 8.39%, O 22.19%.