7183. Perphenazine
Nomenclature
CAS number: 58-39-9
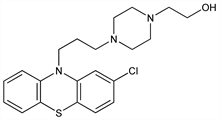
4-[3-(2-Chloro-10H-phenothiazin-10-yl)propyl]-1-piperazineethanol; 2-chloro-10-[3-[1-(2-hydroxyethyl)-4-piperazinyl]propyl]phenothiazine; 1-(2-hydroxyethyl)-4-[3-(2-chloro-10-phenothiazinyl)propyl]piperazine; chlorpiprazine; chlorpiprozine; PZC; Sch-3940; Trilafon (Schering); Trilifan; Decentan (Merck KGaA); Fentazin (Allen & Hanburys); Perphenan. C21H26ClN3OS; mol wt 403.97.
C 62.44%, H 6.49%, Cl 8.78%, N 10.40%, O 3.96%, S 7.94%.
Description and references
Prepn: Cusic, US 2766235 (1956); Sherlock, Sperber, US 2860138 (1958 to Schering). Metabolism: U. Breyer, H. J. Gaertner, Adv. Biochem. Psychopharmacol. 9, 167 (1974); H. J. Gaertner et al., Drug Metab. Dispos. 3, 437 (1975). Crystal structure: J. J. H. McDowell, Acta Crystallogr. B34, 686 (1978).
Properties
Crystals. Sensitive to light. mp 94-100°. bp0.15 214-218°; bp1 278-281°. Practically insol in water. Soly (mg/ml): ethanol 153; acetone 82. Practically insol in sesame oil.Derivative
Dihydrochloride.C21H28Cl3N3OS; mol wt 476.89.
C 52.89%, H 5.92%, Cl 22.30%, N 8.81%, O 3.35%, S 6.72%.
Properties
Crystals from alcohol, mp 225-226°.Therapeutic Category
Antipsychotic.
Therapeutic Category (Veterinary)
Tranquilizer, pre-anesthetic agent.Keywords
Antipsychotic; Phenothiazines