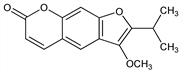
7191. Peucedanin
Nomenclature
CAS number: 133-26-6
3-Methoxy-2-(1-methylethyl)-7H-furo[3,2-g][1]benzopyran-7-one; 6-hydroxy-2-isopropyl-3-methoxy-5-benzofuranacrylic acid δ-lactone; 4-methoxy-5-isopropylfuro[2,3:6,7]coumarin; oreoselone methyl ether. C15H14O4; mol wt 258.27.
C 69.76%, H 5.46%, O 24.78%.
Description and references
Coumarin deriv obtained from rhizome of Peucedanum officinale L., Umbelliferae: Schlatter, Ann. 5, 201 (1833); Hlasiwetz, Weidel, ibid. 174, 67 (1874); A. Jassoy, P. Haensel, Arch. Pharm. 236, 662 (1898); Popper, Monatsh. Chem. 19, 268 (1898); from Peucedanum morisonii, Bess., Umbelliferae: G. K. Nikonov, A. A. Ivashenko, Zh. Obshch. Khim. 33, 2740 (1963). Fluorescence spectrum: R. H. Goodwin, F. Kavanagh, Arch. Biochem. 27, 182 (1950). Antitumor activity and toxicity studies: E. M. Vermel, S. A. Kruglyak-Syrkina, Vopr. Onkol. 5, 43 (1959), C.A. 53, 19162h (1959). Structure: E. Sp"ath et al., Ber. 64, 2203 (1931); E. Sp"ath, K. Klager, ibid. 66, 749 (1933). Synthesis: H. Schmid, A. Ebn"other, Helv. Chim. Acta 34, 1982 (1951).