7218. Phenazocine
Nomenclature
CAS number: 127-35-5
1,2,3,4,5,6-Hexahydro-6,11-dimethyl-3-(2-phenylethyl)-2,6-methano-3-benzazocin-8-ol; 2′-hydroxy-5,9-dimethyl-2-phenethyl-6,7-benzmorphan; phenethylazocine; phenobenzorphan. C22H27NO; mol wt 321.46.
C 82.20%, H 8.47%, N 4.36%, O 4.98%.
Description and references
Synthetic opioid analgesic. Prepn of (±)-cis-form from metazocine, q.v.: E. L. May, N. B. Eddy, J. Org. Chem. 24, 294 (1959). Optical resolution and pharmacology: eidem, ibid. 1435. Improved syntheses: J. H. Ager, E. L. May, ibid. 25, 984 (1960); G. A. Brine et al., J. Heterocycl. Chem. 27, 2139 (1990).
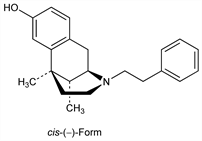
Derivative
cis-(±)-Form.Nomenclature
CAS number: 58073-76-0
(±)-α-Phenazocine. Properties
Rods from methanol, mp 181-182°.Derivative
cis-(±)-Form hydrobromide.Nomenclature
CAS number: 70878-79-4;
1239-04-9
(unspecified stereo)
NIH-7519; Narphen (Napp); Prinadol. C22H27NO.HBr; mol wt 402.37.
C 65.67%, H 7.01%, N 3.48%, O 3.98%, Br 19.86%.
Properties
Rods from acetone or abs alc + ether, mp 166-170°. LD50 s.c. in mice: 332 mg/kg (May, Eddy).Derivative
cis-(-)-Form.Nomenclature
CAS number: 58640-87-2
α-(-)-Phenazocine. Properties
Needles from methanol, mp 159-159.5°. [α]D20 -122° (c = 0.74 in 95% ethanol).Derivative
cis-(-)-Form hydrobromide.Properties
Crystals, mp 284-287°. [α]D20 -84.1° (c = 1.12 in 95% ethanol). LD50 s.c. in mice: 147 mg/kg (May, Eddy).Note
This is a controlled substance (opiate): 21 CFR, 1308.12.Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids; Benzomorphan Derivatives