7239. Phenocoll
Nomenclature
CAS number: 103-97-9
2-Amino-N-(4-ethoxyphenyl)acetamide; 2-amino-p-acetophenetidide; α-amino-p-acetophenetide; aminoacetophenetidine; 4-glycylaminophenol ethyl ether; glycine p-phenetidide; 4-glycylaminophenetol; glycocoll-p-phenetidide; phenokoll. C10H14N2O2; mol wt 194.23.
C 61.84%, H 7.27%, N 14.42%, O 16.47%.
Description and references
Prepn: Majert, DE 59121; DE 59874 (1891 to Schering), Frdl. 3, 915, 918; DE 346809 (1921 to Schering), Frdl. 13, 1066; Karrer, Haebler, Helv. Chim. Acta 7, 534 (1924).
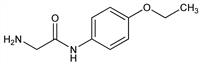
Properties
Crystals, mp 100.5°.Derivative
Monohydrate.Properties
Crystals, mp 95°.Derivative
Hydrochloride.Nomenclature
CAS number: 539-10-6
C10H14N2O2.HCl; mol wt 230.69.
C 52.06%, H 6.55%, N 12.14%, O 13.87%, Cl 15.37%.
Properties
Cryst powder. Sol in 20 parts water, in alcohol. More sol in hot water; slightly sol in benzene, chloroform, ether. Aq soln is neutral. Incompat. Alkali hydroxides or carbonates.Derivative
Salicylate.Nomenclature
CAS number: 140-47-6
Salocoll. C10H14N2O2.C7H6O3; mol wt 332.35.
C 61.44%, H 6.07%, N 8.43%, O 24.07%.
Properties
Fine needles. Sweetish taste. Sol in 200 parts cold, 20 parts hot water. Incompat. As for the hydrochloride; with substances incompat with salicylates.Therapeutic Category
Antipyretic, analgesic.