7241. Phenol
Nomenclature
Description and references
Obtained from coal tar, or made by fusing sodium benzenesulfonate with NaOH, or by heating monochlorobenzene with aq NaOH under high pressure. The crystalline article of commerce contains at least 98% phenol. Review of mfg processes: A. Dierichs, R. Kubicka, Phenole und Basen, Vorkommen und Gewinnung (Akademie-Verlag, Berlin, 1958) 472 pp; Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 612-623. Use in treatment of spasticity: D. E. Garland et al., Clin. Orthop. 165, 217 (1982); eidem, Arch. Phys. Med. Rehabil. 65, 243 (1984). Toxicity study: W. B. Deichmann, S. Witherup, J. Pharmacol. Exp. Ther. 80, 233 (1944). Review of use in pain relief: K. M. Wood, Pain 5, 205-229 (1978). Review: C. Thurman in Kirk-Othmer Encyclopedia of Chemical Technology vol. 17 (Wiley-Interscience, New York, 3rd ed., 1982) pp 373-384. Review of toxicology: H. Babich, D. L. Davis, Regul. Toxicol. Pharmacol. 1, 90-109 (1981); and human exposure: Toxicological Profile for Phenol (PB99-122012, 1998) 241 pp.
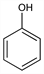
Properties
Colorless, acicular crystals or white, crystalline mass. Characteristic odor, somewhat sickeningly sweet and acrid with a sharp and burning taste. Poisonous and caustic. Prone to redden on exposure to air and light, hastened by presence of alkalinity. d 1.071. When free from water and cresols it congeals at 41° and melts at 43°. Ultrapure material mp 40.85°. The commercial product contains an impurity which raises the mp. bp 182°. Flash pt, closed cup: 175°F (79°C). nD41 1.5425. pKa at 25° = 10.0. pH of aq solns ≈6.0. It is liquefied by mixing with ≈8% water. One gram dissolves in ≈15 ml water, 12 ml benzene; very sol in alcohol, chloroform, ether, glycerol, carbon disulfide, petrolatum, volatile and fixed oils, aq alkali hydroxides. Almost insol in petr ether. LD50 orally in rats: 530 mg/kg (Deichmann, Witherup). Keep well closed and protected from light. Do not handle with bare hands.Derivative
Ammonium salt.Nomenclature
Properties
White to pink crystalline masses. Sol in water.Derivative
Sodium salt.Nomenclature
Description and references
Crystal structure and reactivity study: M. Kunert et al., Ber./Recl. 130, 1461 (1997).
Properties
Colorless crystals, mp 380°. Very sol in water; sol in alcohol. The aq soln is caustic.Caution
Potential symptoms of acute overexposure are steatorous breathing, mucous rales, froth at mouth and nose, frank pulmonary edema; cyanosis; tremor, convulsions, twitching; death due to respiratory failure. Chronic overexposure may result in vomiting, difficulty swallowing, excess salivation, diarrhea, anorexia, weight loss; headache, fainting, vertigo, mental disturbances; muscle aches and pain, weakness; damage to liver and kidney, dark urine. Ingestion may cause burning of mouth and throat; white necrotic lesions in mouth, esophagus and stomach; abdominal pain. Direct contact may cause irritation of eyes, nose and throat; skin burns; dermatitis; ochronosis. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 2003-100121, 2003) p 248; Clinical Toxicology of Commercial Products, R. E. Gosselin et al., Eds. (Williams & Wilkins, Baltimore, 5th ed., 1984) Section III, p344-348; Patty's Industrial Hygiene and Toxicology vol. 2B, G. D. Clayton, F. E. Clayton, Eds. (Wiley-Interscience, New York, 4th ed., 1994) p 1567-1584.Use
As a general disinfectant, either in soln or mixed with slaked lime, etc., for toilets, stables, cesspools, floors, drains, etc.; for the manuf of colorless or light-colored artificial resins, many medical and industrial organic compds and dyes; as a reagent in chemical analysis. pH indicator. Pharmaceutic aid (preservative).Therapeutic Category
Aqueous soln as topical anesthetic; topical antiseptic; topical antipruritic.
Therapeutic Category (Veterinary)
Antiseptic caustic. Topical anesthetic in pruritic skin conditions. Has been used internally and externally as an antiseptic.Keywords
Anesthetic (Local); Antipruritic