Nomenclature
CAS number: 108-45-2
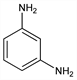
1,3-Benzenediamine;
m-diaminobenzene.
C
6H
8N
2; mol wt 108.14.
C 66.64%, H 7.46%, N 25.90%.
Description and references
Prepd by the reduction of m-dinitrobenzene:
Kuhn, US 2768209 (1956 to Ringwood); Faust, J. Prakt.
Chem. 6, 14 (1958); Neilson et al., J. Chem. Soc. 1962, 371; Tallee, Peltier, Compt. Rend. 259, 400 (1964). Toxicity study: C. Burnett et
al., J. Toxicol. Environ. Health 2, 657 (1977).
Properties
White crystals becoming red on exposure to air.
d 1.139; mp 62-63°; bp 284-287°; dipole moment 1.79. Fire point:
175°. Sol in water, methanol, ethanol, chloroform, acetone, dimethylformamide,
methyl ethyl ketone, dioxane. Slightly sol in ether, carbon tetrachloride,
dibutyl phthalate, isopropanol. Very slightly sol in benzene, toluene,
xylene, butanol. Keep well closed and protected
from light. LD50 in rats
(mg/kg): 650 orally; 283 i.p. (Burnett).Derivative
Hydrochloride.
Properties
White or slightly red, crystalline powder; becomes
darker on exposure to air. Freely sol in water; sol in alcohol.Use
Manuf dyes; rubber curing agents, ion exchange resins,
decolorizing resins, formaldehyde condensates, resinous polyamides,
block polymers, textile fibers, urethanes, petroleum additives, rubber
chemicals, corrosion inhibitors; in photography; as reagent for gold
and bromine. The hydrochloride chiefly as a reagent for nitrite.
