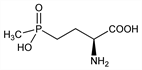
7339. Phosphinothricin
Nomenclature
Description and references
First natural l-amino acid containing a phosphinic acid group. Isolated from the tripeptide antibiotic, bialaphos; inhibits glutamine synthetase. Isoln from S. viridochromogenes and synthesis of dl-form: E. Bayer et al., Helv. Chim. Acta 55, 224 (1972). Synthesis of dl-form: H. Gross, T. Gnauk, J. Prakt. Chem. 318, 157 (1976); of enantiomers: N. Minowa et al., Tetrahedron Lett. 25, 1147 (1984). Crystal structure: E. F. Paulus, S. Grabley, Z. Kristallogr. 160, 63 (1982). Determn in biological fluids: A. Suzuki, M. Kawana, Bull. Environ. Contam. Toxicol. 43, 17 (1989). Environmental impact in lakes: M. J. Faber et al., Environ. Toxicol. Chem. 17, 1291 (1998). Field trial in orchards and vineyards as herbicide: P. Langelüddeke et al., Meded. Fac. Landbouwwet. Rijksuniv. Gent 47, 95 (1982); on seed potato as desiccant: H. M. Lawson, J. S. Wiseman, Brighton Crop Prot. Conf. - Weeds 1991, 233. Review of syntheses, activities and uses: G. Hoerlein, Rev. Environ. Contam. Toxicol. 138, 73-145 (1994).