Nomenclature
CAS number: 7664-38-2
Orthophosphoric acid.
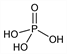
H
3O
4P; mol wt 98.00.
H 3.09%, O 65.30%, P 31.61%.
H
3PO
4.
Description and references
Obtained commercially from phosphate rock deposits
in Florida, Tennessee, and the Western United States. Phosphate rock
is essentially tricalcium phosphate and one of the large scale processes
is based on the equation: Ca3(PO4)2 + 3H2SO4 + 6H2O → 2H3PO4 + 3(CaSO4.2H2O). Description
of various processes: W. H. Waggaman, Phosphoric
Acid, Phosphates and Phosphatic Fertilizers (Reinhold,
New York, 1952); Phosphoric Acid Vol. 1, parts I, II, A. V. Slack, Ed. (Dekker, New York,
1968) 1159 pp; Faith, Keyes & Clark's
Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds.
(Wiley-Interscience, New York, 4th ed., 1975) pp 628-639. Prepn of
ultrapure, cryst H3PO4: Simon, Schulze, Z. Anorg. Allg. Chem. 242, 322
(1939); Weber, King, Inorg. Synth. 1, 101 (1939). Reviews: J. R. Van Wazer, Phosphorus and Its Compounds Vol. 1, Chemistry (Interscience, New
York, 1958) pp 479-491; R. B. Hudson et al., “Phosphoric
Acids and Phosphates” in Kirk-Othmer Encyclopedia
of Chemical Technology Vol. 17 (Wiley-Interscience,
New York, 3rd ed., 1982) pp 426-472.
Properties
Unstable, orthorhombic crystals, mp 42.35°, or clear, syrupy liquid;
easily supercooled into a glass. Pleasing acid taste when suitably
diluted. An acid containing ≈88% H3PO4 will
frequently crystallize on prolonged cooling; forms hemihydrate, mp 29.32°. Becomes anhydr
at 150°, gradually changes to pyrophosphoric acid at ≈200°, and changes
to metaphosphoric acid when heated above 300°. The hot concd acid
attacks porcelain and granite ware. May be stored in suitable stainless
steel containers. Tribasic acid: pK1: 2.15; pK2: 7.09; pK3: 12.32. Other reported values of dissociation constants and viscosity
data reviewed by Van Wazer. The pH of a 0.1N aq soln is
1.5. Heat of formation (crystals): -306.2 kcal/mole. Heat of soln:
+2.79 kcal/mole. Corrosive.
Sol in water, alc; sol in 8 vols of a 3:1 ether: alcohol mixture.
Properties of phosphoric acid solns. d25 1.8741 (100% soln); 1.6850 (85% soln); 1.3334 (50% soln); 1.0523 (10% soln). Density measurements:
Christensen, Reed, Ind. Eng. Chem. 47, 1277 (1950); Egan, Luff, ibid. 1280. nD17.5 1.34203 (10% soln); 1.35032 (20% soln); 1.35846 (30% soln). Spec heat (21.3°): 0.4359 (88% soln).Caution
Potential symptoms of overexposure
are irritation of upper respiratory system, eyes and skin; burns skin
and eyes; dermatitis. See NIOSH
Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997)
p 254.Use
In the manuf of superphosphates for fertilizers,
other phosphate salts, polyphosphates, detergents. Acid catalyst
in making ethylene, purifying hydrogen peroxide. As acidulant and
flavor, synergistic antioxidant and sequestrant in food. Pharmaceutic
aid (solvent). In dental cements; process engraving; rustproofing
of metals before painting; coagulating rubber latex; as analytical
reagent for trace metal analytsis. In buffers.
Therapeutic Category (Veterinary)
Has been used in lead poisoning.
