7384. Physostigmine
Nomenclature
Description and references
Acetylcholinesterase inhibitor. From Calabar beans, the seeds of the vine Physostigma venenosum Balf., Leguminosae: Jobst, Hesse, Ann. 129, 115 (1864); Hesse, ibid. 141, 82 (1867). Extraction procedure: Schwyzer, Die Fabrikation pharmazeutischer und chemisch-technischer Produkte (Berlin, 1931) p 338; Chemnitius, J. Prakt. Chem. 116, 59 (1927). Structure: Stedman, Barger, J. Chem. Soc. 127, 247 (1925). Absolute configuration: R. B. Longmore, B. Robinson, Chem. Ind. (London) 1969, 622. Crystal structure: Petcher, Pauling, Nature 241, 277 (1973). Synthesis: Julian, Pikl, J. Am. Chem. Soc. 57, 755 (1935); Harley-Mason, Jackson, J. Chem. Soc. 1954, 3651; J. Wijnberg, W. N. Speckamp, Tetrahedron 34, 2399 (1978). Cholinesterase inhibitor: Engelhart, Loewi, Arch. Exp. Pathol. Pharmakol. 150, 1 (1930); Matthes, J. Physiol. 70, 338 (1930). Toxicity data: W. T. Lynch, J. M. Coon, Toxicol. Appl. Pharmacol. 21, 53 (1972); R D. Sofia, L. C. Knabloch, ibid. 28, 227 (1974). Improvement of long-term memory: K. L. Davis et al., Science 201, 272 (1978). Memory enhancement in Alzheimer's disease: L. J. Thal et al., N. Engl. J. Med. 308, 720 (1983); eidem, Ann. Neurol. 13, 491 (1983); L. Gustafson et al., Psychopharmacology 93, 31 (1987). Review: B. Robinson in The Alkaloids Vol. XIII, R. H. F. Manske, Ed. (Academic Press, New York, 1971) pp 213-226. Comprehensive description: F. J. Muhtadi, S. S. El-Hawary, Anal. Profiles Drug Subs. 18, 289-350 (1989).
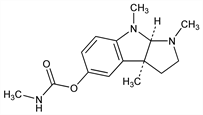
Properties
Orthorhombic sphenoidal prisms or clusters of leaflets from ether or benzene. mp 105-106° (also an unstable, low-melting form, mp 86-87°). [α]D17 -76° (c = 1.3 in chloroform); [α]D25 -120° (benzene). pKa1 6.12; pKa2 12.24. Slightly sol in water; sol in alc, benzene, chloroform, oils. Solid and solns turn red on exposure to heat, light, air, and on contact with traces of metals. Under certain conditions the oxidation may proceed to yield eserine blue, C26H31N5O2 . LD50 orally in mice: 4.5 mg/kg (Lynch, Coon).Derivative
Salicylate.Nomenclature
Properties
Acicular crystals, mp 185-187°. uv max (methanol): 239, 252, 303 nm (log ε 4.09, 4.04, 3.78). One gram dissolves in 75 ml water at 25° (pH of 0.5% aq soln 5.8); in 16 ml water at 80°; in 16 ml alcohol, 5 ml boiling alcohol, in 6 ml chloroform, 250 ml ether. Solns should be kept well closed in light-resistant, alkali-free glass containers and used within a week. Turns red and loses effectiveness on exposure to heat, light, air. LD50 i.p. in mice: 0.64 mg/kg (Sofia, Knabloch).Derivative
Sulfate.Nomenclature
Properties
Deliquescent scales, mp 140° (after drying at 100°). One gram dissolves in 0.4 ml alcohol, 4 ml water (pH of 0.05M soln 4.7), 1200 ml ether. The solns are more prone to change color than those of the salicylate.Derivative
Sulfite.Properties
White powder, freely sol in water, alcohol. The aq soln is stated to remain colorless for a long time.Therapeutic Category
Cholinergic; miotic.
Therapeutic Category (Veterinary)
Cholinergic; miotic.Keywords
Cholinergic; Miotic