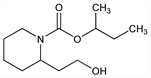
7394. Picaridin
Nomenclature
CAS number: 119515-38-7
2-(2-Hydroxyethyl)-1-piperidinecarboxylic acid 1-methylpropyl
ester; 1-methylpropyl 2-(2-hydroxyethyl)piperidine-1-carboxylate; hydroxyethyl isobutyl piperidine carboxylate; icaridin; KBR 3023; Bayrepel (Bayer); Saltidin (Saltigo). C12H23NO3; mol wt 229.32.
C 62.85%, H 10.11%, N 6.11%, O 20.93%.
Description and references
Acts on certain olfactory receptor cell types to reduce the activating or attracting effect of odor sources. Prepn: B.-W. Krüger et al., EP 289842; eidem, US 4900834 (1988, 1990 both to Bayer). Synthesis and mode of action: J. Boeckh et al., Pestic. Sci. 48, 359 (1996) DOI. Field trials in mosquito repellency: H. H. Yap et al., J. Vector Ecol. 23, 62 (1998) PubMed; A. Badolo et al., Trop. Med. Int. Health 9, 330 (2004) DOI PubMed. Physical, chemical and toxicological properties: WHO Interim Specification for Icaridin (WHO/IS/TC/ 668/2001) 16 pp. MS characterization: T. P. Knepper, J. Chromatogr. A 1046, 159 (2004) DOI PubMed.