Nomenclature
CAS number: 88-89-1
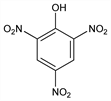
2,4,6-Trinitrophenol; 1-hydroxy-2,4,6-trinitrobenzene; picronitric acid; carbazotic acid; nitroxanthic acid.
C
6H
3N
3O
7; mol wt 229.10.
C 31.46%, H 1.32%, N 18.34%, O 48.89%.
Description and references
Prepd by sulfonating phenol then treating with
nitric acid: Olsen, Goldstein, Ind. Eng.
Chem. 16, 66 (1924); by treating benzene with
nitric acid and mercuric nitrate: Teeters, Mueller, US 2455322 (1948 to Allied Chem.); by nitration of 2-tert-butyl-4,6-dinitropenol:
Ley, Müller, Ber. 89, 1402 (1956). Crystal structure: E. N. Duesler et al., Cryst. Struct. Commun. 7, 449 (1978).
Properties
Pale yellow, odorless, intensely bitter crystals.
d 1.763. mp 122-123°. Explodes above 300°.
One gram dissolves in 78 ml water, 15 ml boiling water, 12 ml alc,
10 ml benzene, 35 ml chloroform, 65 ml ether. Keep in a cool place and remote from fire. Explodes when rapidly heated or by percussion. Note: For safety in transportation, 10-20% water is
usually added.Caution
Potential symptoms of overexposure
are irritation of eyes, skin; sensitization dermatitis; yellow stained
hair, skin; weakness, myalgia, anuria, polyuria; bitter taste, GI
disturbances; hepatitis; hematuria, albuminuria, nephritis. See NIOSH Pocket Guide to Chemical
Hazards (DHHS/NIOSH 97-140, 1997) p 258; Clinical Toxicology of Commercial Products, R. E. Gosselin et al., Eds. (Williams & Wilkins,
Baltimore, 5th ed., 1984) Section II, p 197.Use
Explosives, matches; in leather industry; electric
batteries; etching copper; manuf colored glass; textile mordant.
Reagent for prepn of organic derivatives for identification.
