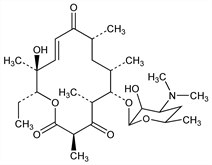
7413. Picromycin
Nomenclature
CAS number: 19721-56-3
(E)-14-Ethyl-13-hydroxy-3,5,7,9,13-pentamethyl-6-[[3,4,6-trideoxy-3-(dimethylamino)-β-d-xylo-hexopyranoside]oxy]oxacyclotetradec-11-ene-2,4,10-trione; pikromycin; albomycetin; amaromycin. C28H47NO8; mol wt 525.67.
C 63.98%, H 9.01%, N 2.66%, O 24.35%.
Description and references
First macrolide antibiotic isolated. Isoln from Actinomyces spp by Lindenbein and Bauer, see Brockmann, Henkel, Naturwissenschaften 37, 138 (1950); Ber. 84, 284 (1951); Brockmann, Bohne, US 2693433 (1954 to Schenley). Structure: Brockmann, Oster, Ber. 90, 605 (1957); Anliker, Gubler, Helv. Chim. Acta 40, 119, 1768 (1957). Studies in stereochemistry: Djerassi, Halpern, J. Am. Chem. Soc. 79, 3926 (1957); Ogura et al., ibid. 97, 1930 (1975); eidem, Tetrahedron 37, Suppl. 1, 165 (1981). Revised structure: Rickards, Smith, Chem. Commun. 1968, 1049; Muxfeldt et al., J. Am. Chem. Soc. 90, 4748 (1968). Synthesis from narbonolide: Maezawa et al., J. Antibiot. 26, 771 (1973).