7424. Pilocarpine
Nomenclature
Description and references
Cholinergic principle from Pilocarpus jaborandi Holmes, Rutaceae. Isoln: Petit, Polanovski, Bull. Soc. Chim. [3] 17, 557, 702 (1897). Structure: Jowett, J. Chem. Soc. 77, 473, 851 (1900); 83, 438 (1903). Stereoisomeric with isopilocarpine: Polonovski, Polonovski, Bull. Soc. Chim. [4] 31, 1314 (1922). Has the cis configuration; isopilocarpine is trans: Zav'yalov, Dokl. Akad. Nauk SSSR 82, 257 (1952). Absolute configuration: Hill, Barcza, Tetrahedron 22, 2889 (1966). Synthesis: Preobrashenski et al., Ber. 66, 1187 (1933); Samokhvalov, Med. Prom. SSSR 11, no. 2, 10 (1957); DeGraw, Tetrahedron 28, 967 (1972); Link, Bernauer, Helv. Chim. Acta 55, 1053 (1972). Stereoselective synthesis: A. Noordam et al., Rec. Trav. Chim. 98, 467 (1979). Review: Langenbeck, Angew. Chem. 60, 297 (1948); van Rossum et al., Experientia 16, 373 (1960). Toxicity studies: Beccari, Boll. Chim. Farm. 106, 8 (1967). Comprehensive description: A. A. Al-Badr, H. Y. Aboul-Enein, Anal. Profiles Drug Subs. 12, 385-432 (1983). Clinical trial in Sj"ogren's syndrome: F. B. Vivino et al., Arch. Intern. Med. 159, 174 (1999); in radiation-induced xerostomia: J.-C. Horiot et al., Radiother. Oncol. 55, 233 (2000).
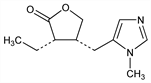
Properties
Oil or crystals, mp 34°. bp5 260° (partial conversion to isopilocarpine). [α]D18 +106° (c = 2). pK1 (20°) 7.15; pK2 (20°) 12.57. Sol in water, alcohol, chloroform; sparingly sol in ether, benzene. Almost insol in petr ether.Derivative
Hydrochloride.Nomenclature
Properties
Hygroscopic crystals from alcohol, mp 204-205°. [α]D18 +91° (c = 2). Freely sol in water, alcohol. Practically insol in ether, chloroform. Keep well closed and protected from light.Derivative
Nitrate.Nomenclature
Properties
mp 173.5-174.0° (dec). Poisonous. [α]D +77 to +83° (c = 10). One gram dissolves in 4 ml water, 75 ml alcohol. Insol in chloroform, ether. Incompat. Silver nitrate, mercury bichloride, iodides, gold salts, tannin, calomel, KMnO4, alkalies.Derivative
Isopilocarpine.Nomenclature
β-Pilocarpine.Properties
Hygroscopic oily liquid or prisms. bp10 261°. [α]D18 +50° (c = 2). pK1 (18°) 7.17. Miscible with water and alcohol; very sol in chloroform; less sol in benzene, ether. Almost insol in petr ether.Derivative
Isopilocarpine hydrochloride hemihydrate.Properties
Scales from alcohol + ether, mp 127°; when anhydr, mp 161°. [α]D18 +39° (c = 5). Sol in 0.27 part water; 2.1 parts alcohol.Derivative
Isopilocarpine nitrate.Properties
Prisms from water, scales from alcohol, mp 159°. [α]D18 +39° (c = 2). Sol in 8.4 parts water, in 350 parts abs alcohol.Therapeutic Category
Antiglaucoma agent; miotic; sialogogue.
Therapeutic Category (Veterinary)
Parasympathomimetic; miotic; gastric secretory stimulant.Keywords
Antiglaucoma; Miotic; Sialagogue