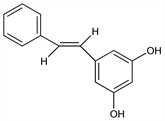
7451. Pinosylvin
Nomenclature
Description and references
Occurs together with its monomethyl and dimethyl ethers in the heartwood of pine and other woody plants. Naturally occurring pinosylvins have the trans configuration. Isoln from Pinus sylvestris L., Pinaceae: H. Erdtman, Ann. 539, 116 (1939); from other Pinus species: G. Lindstedt, Acta Chem. Scand. 3, 755-772 (1949); J. C. Alvarez-Novoa et al., ibid. 4, 444 (1950); from Alnus sieboldiana, Betulaceae: Y. Asakawa, Bull. Chem. Soc. Jpn. 44, 2761 (1971); from Polygonum nodosum, Polygonaceae: M. Kuroyanagi et al., Chem. Pharm. Bull. 30, 1602 (1982). Synthesis of pinosylvin: E. Sp"ath, F. Liebherr, Ber. 74, 869 (1941); of monomethyl ether: E. Sp"ath, K. Kromp, ibid. 1424; of dimethyl ether: G. Aulin-Erdtman, H. Erdtman, ibid. 50; of pinosylvin and derivatives: A. A. Loman, L. R. Snowdon, Can. J. Chem. 48, 1554 (1970). Biosynthesis: Birch, Fortschr. Chem. Org. Naturst. 14, 186 (1957). Toxicological study: K. O. Frykholm, Nature 155, 454 (1945). Use as antimicrobial agent: E. H. Sheers, DE 1952451; idem, US 3577230 (1970, 1971 both to Arizona Chem. Co.). Deterrent to feeding behavior of snowshoe hare: J. P. Bryant et al., Science 222, 1023 (1983).