7468. Piperidine
Nomenclature
CAS number: 110-89-4
Hexahydropyridine. C5H11N; mol wt 85.15.
C 70.53%, H 13.02%, N 16.45%.
Description and references
Found in small quantities in Piper nigrum L., Piperaceae (black pepper). May be obtained from piperine by heating with alcoholic KOH, or from 1,5-diaminopentane hydrochloride by cyclization. Usually prepd by electrolytic reduction of pyridine. Forms complexes with salts of heavy metals. Because of its reactivity, piperidine is useful in the prepn of cryst derivatives of aromatic nitro compds contg nuclear halogen atoms: Seikel, J. Am. Chem. Soc. 62, 750 (1940). Toxicity study: H. F. Smyth et al., Am. Ind. Hyg. Assoc. J. 23, 95 (1962). Review of physical constants of piperidine and N-alkyl piperidines: Magnusson, Schierz, Univ. Wyo. Publ. 7, 1 (1940).
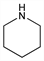
Properties
Liquid. Characteristic odor. Soapy feel. Solidifies -13° to -17°; mp -7°; bp760 106°; bp20 18°; d420 0.8622; nD20 1.4534. Infrared absorption spectrum: Freymann, Compt. Rend. 205, 852 (1937); Ann. Chim. 11, 11 (1939). Ultraviolet and Raman spectra: Lecomte, Compt. Rend. 207, 395 (1938). Corrosive; flammable. Strong base: pK (25°): 2.80. Misc with water. Sol in alcohol, benzene, chloroform. LD50 orally in rats: 0.52 ml/kg (Smyth).Derivative
Hydrochloride.C5H11N.HCl; mol wt 121.61.
C 49.38%, H 9.95%, N 11.52%, Cl 29.15%.
Properties
Orthorhombic prisms from alcohol, mp 247°. Freely sol in water, alcohol.Derivative
Nitrate.C5H11N.HNO3; mol wt 148.16.
C 40.53%, H 8.16%, N 18.91%, O 32.40%.
Properties
Hygroscopic plates, sublimes 75° (10 mm), mp 110°. Freely sol in water, alcohol, ether. Absorption spectrum: Harper, Macbech, J. Chem. Soc. 107, 91 (1915).Derivative
Bitartrate.C5H11N.C4H6O6; mol wt 235.23.
C 45.95%, H 7.28%, N 5.95%, O 40.81%.
Properties
Crystals, freely sol in water.Derivative
Aurichloride.C5H11N.HAuCl4; mol wt 424.93.
C 14.13%, H 2.85%, N 3.30%, Au 46.35%, Cl 33.37%.
Properties
Yellow crystals, mp 219°.Derivative
Platinichloride.2C5H11N.H2PtCl6; mol wt 580.11.
C 20.70%, H 4.17%, N 4.83%, Pt 33.63%, Cl 36.67%.
Properties
Yellow monoclinic prisms from water, mp 202°. Freely sol in water, slightly in alcohol.Derivative
N-Benzoylpiperidine.Properties
Long needles, mp 44-48°. Ref: Org. Synth. coll. vol. I (2nd ed., 1941) p 99.Derivative
Phosphate.Nomenclature
CAS number: 767-21-5
Description and references
Prepn from piperidine + phosphoric acid: Abood, US 3035977 (1962).