7488. Pirarubicin
Nomenclature
Description and references
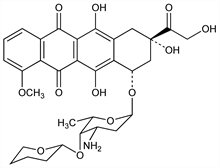
Structural analog of doxorubicin, q.v. Prepn of (2″R) and (2″S)-diastereomers: H. Umezawa et al., J. Antibiot. 32, 1082 (1979); idem et al., EP 14853; eidem, US 4303785 (1980, 1981 both to Microbiochem. Res. Found. Japan). Absolute configuration: idem et al., J. Antibiot. 37, 1094 (1984). HPLC determn in biological fluids: Y. Matsushita et al., ibid. 36, 880 (1983). Cellular uptake and inhibition of DNA synthesis: S. Kunimoto et al., ibid. 312 (1983). Mechanism of action study: K.-I. Kiyomiya et al., Int. J. Oncol. 21, 1081 (2002). Clinical pharmacokinetics and toxicity: A. A. Miller, C. G. Schmidt, Cancer Res. 47, 1461 (1987). Overview of clinical experience: H. Majima, K. Ohta, Biomed. Pharmacother. 41, 237-243 (1987). Clinical evaluation in metastatic colon cancer: D. Fallik et al., Ann. Oncol. 14, 856 (2003)
Properties
Red solid, mp 188-192° (dec). [α]D25 +175 ±25° (c = 0.2 in CHCl3). uv and visible max (methanol): 234, 252, 290, 498, 531.5, 580 nm (E1%1cm 480, 350, 110, 140, 100, 45). Sol in ethyl acetate, chloroform, and ethanol; slightly sol in water, n-hexane, petr ether. Ethanolic and acidic solutions are red in color; give a positive ninhydrin reaction and do not reduce Fehling's solution. LD50 i.v. in mice: 27.8 mg/kg (Umezawa, 1979).Derivative
Hydrochloride.Nomenclature
Properties
Red crystalline solid. Sol in water and methanol.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Antibiotics and Analogs; Anthracyclines; Topoisomerase II Inhibitor