7492. Piretanide
Nomenclature
CAS number: 55837-27-9
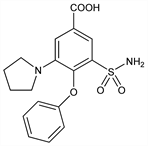
3-(Aminosulfonyl)-4-phenoxy-5-(1-pyrrolidinyl)benzoic acid; 4-phenoxy-3-(1-pyrrolidinyl)-5-sulfamoylbenzoic acid; HOE-118; S-73-4118; Diumax (Cusi-Norte); Eurelix (Hoechst); Tauliz (HMR). C17H18N2O5S; mol wt 362.40.
C 56.34%, H 5.01%, N 7.73%, O 22.07%, S 8.85%.
Description and references
High-ceiling loop diuretic, structurally related to bumetanide, q.v. Prepn: D. Bormann et al., DE 2419970; eidem, US 4010273 (1975, 1977 both to Hoechst). Chemistry and pharmacology: W. Merkel et al., Eur. J. Med. Chem. 11, 399 (1976). Diuretic activity in man: N. Pozet et al., Br. J. Clin. Pharmacol. 9, 577 (1980); T. Saruta, E. Kato, Arzneim.-Forsch. 30, 1807 (1980). Vascular effects of piretanide: E. Klaus et al., ibid. 33, 1273 (1983); renal effects: M. Omosu et al., ibid. 1277. Review of pharmacology and therapeutic efficacy: S. P. Clissold, R. N. Brogden, Drugs 29, 489-530 (1985).
Properties
Pale yellow platelets from methanol/water, mp 225-227°. Exhibits intense light blue fluorescence at 366 nm. LD50 in rats, mice (mg/kg): 5601, 3672 orally (Merkel).Derivative
Monosodium salt.Nomenclature
CAS number: 112132-09-9
Arelix (HMR). C17H17N2NaO5S; mol wt 384.38.
C 53.12%, H 4.46%, N 7.29%, Na 5.98%, O 20.81%, S 8.34%.
Therapeutic Category
Diuretic.
Keywords
Diuretic; Sulfonamide Derivatives