7545. Podophyllic Acids
C22H24O9; mol wt 432.42.
C 61.11%, H 5.59%, O 33.30%.
Description and references
Since the prepn of a “podophyllic acid”, mp 145-150° (crude), 163-165° (pure), by Borsche, Niemann, Ann. 494, 126 (1932), studies have been made of two isomers: the 2,3-trans-hydroxy acid and the 2,3-cis-hydroxy acid. Prepn of the trans-acid: Kuhn, Wartburg, Experientia 19, 391 (1963); NL 6405480 (1964 to Sandoz), C.A. 62, 9083c (1965); and cis-acid: Renz et al., Ann. 681, 207 (1965). Prepn of a dl-stereoisomer: Gensler et al., J. Am. Chem. Soc. 76, 315 (1954). Configuration and nomenclature: Rutschmann, Renz, Helv. Chim. Acta 42, 890 (1959).
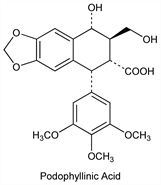
Derivative
2,3-trans-Hydroxy acid.Nomenclature
CAS number: 1853-37-8
Podophyllinic acid. Properties
Crystals from acetone + ether, mp 164-168°. [α]D20 -199.9° (c = 0.463 in ethanol), -292.6° (c = 0.671 in pyridine).Derivative
2,3-cis-Hydroxy acid.Nomenclature
CAS number: 477-67-8
Picropodophyllic acid. Properties
Needles from methanol + ether, double mp 150-155° and 200-232°. [α]D21 -100.4° (c = 0.615 in ethanol), -185° (c = 0.746 in pyridine).Derivative
2,3-trans-Hydroxy acid hydrazide.Nomenclature
CAS number: 78178-41-3
Podophyllinic acid hydrazide. Description and references
Prepn and properties: Rutschmann, US 2977359 (1961 to Sandoz).
Properties
Prisms from methanol, mp 198-199°. [α]D -202° (c = 0.4 in ethanol).Derivative
trans-2-Ethylhydrazide.Nomenclature
CAS number: 1508-45-8
Podophyllinic acid 2-ethylhydrazide; SP-I. Description and references
Prepn: Rutschmann, US 3054802 (1962 to Sandoz).