Nomenclature
PCBs; chlorinated biphenyls; chlorobiphenyls; Aroclor (Monsanto); Clophen (Bayer); Fenclor (Caffaro); Kanechlor (Kanegafuchi); Pyralene (Prodelec). Description and references
Once widely used industrial chemicals whose
high stability contributed to both their commercial usefulness and
their long-term deleterious environmental and health effects. Synthesis:
H. Schmidt, G. Schulz, Ann. 207, 338 (1881). Commercially available since 1930: C. Penning, Ind. Eng. Chem. 22, 1180 (1930).
Commercial PCBs are mixtures of various isomers and congeners. The
Aroclors are characterized by four digit numbers. The first two digits
indicate that the mixture contains biphenyls (12), triphenyls (54)
or both (25, 44); the last two digits give the weight percent of chlorine
in the mixture (e.g. Aroclor 1242 contains biphenyls with ≈42% chlorine).
Accumulation of airborne PCBs in foliage: E. H. Buckley, Science 216, 520 (1982). Reviews: H. L. Hubbard in Kirk-Othmer
Encyclopedia of Chemical Technology vol. 5 (Interscience,
New York, 2nd ed., 1964) pp 289-297; O. Hutzinger et al., The Chemistry of PCBs (CRC Press,
Cleveland, Ohio, 1974) 269 pp; J. W. Lloyd et al., J. Occup. Med. 18, 109-113 (1976).
Reviews of environmental impact and toxicity: L. Fishbein, Annu. Rev. Pharmacol. 14, 139-156
(1974); National Conference on Polychlorinated
Biphenyls, Nov. 19-21, 1975 (EPA-560/6-75-004, 1976) 487
pp; R. D. Kimbrough, Crit. Rev. Toxicol. 2, 445-498 (1974); S. H. Safe, ibid. 24, 87-149 (1994). Reviews of carcinogenicity:
IARC Monographs 18, 43-103 (1978); E. M. Silberhorn et al., Crit. Rev. Toxicol. 20, 439-496
(1990); of toxicology and mechanism of action: S. Safe, ibid. 13, 319-395 (1984); of toxicology
and human exposure: Toxicological Profile
for Polychlorinated Biphenyls (PB2000-108027, 2000) 948
pp.
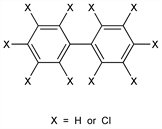
Derivative
Aroclor 1242.
Properties
Clear, mobile liquid; av. number Cl/molecule:
3.10. d425 1.381, d415.5 1.392. Distillation range 325-366°. Flash point (open cup) 348-356°F. nD20 1.627-1.629. Dielectric constant (1000 cycles) 5.6 (25°), 4.9 (100°).Derivative
Aroclor 1254.
Properties
Light yellow, viscous liquid; av. number Cl/molecule:
4.96. d465 1.495; d415.5 1.505. Distillation range 365-390°. No open
cup flash point to boiling. nD20 1.629-1.641. Dielectric
constant (1000 cycles) 5.0 (25°), 4.3 (100°). LD50 orally in weanling rats: 1295 mg/kg (Kimbrough).Derivative
Aroclor 1260.
Properties
Light yellow, soft, sticky resin; av. number Cl/molecule:
6.30. d490 1.555; d415.5 1.566. Distillation range 385-420. No open
cup flash point to boiling. nD20 1.647-1.649. Dielectric
constant (1000 cycles) 4.3 (25°); 3.7 (100°). LD50 orally in weanling rats: 1315 mg/kg (Kimbrough).Caution
In Japan, 1968, oral intoxication
to humans due to accidental contamination of rice bran oil with Kanechlor
400 led to an outbreak of what became known as “Yusho disease”. Symptoms
of oral intoxication in humans included nausea, lethargy, chloracne,
brown pigmentation of skin and nails, subcutaneous edema of the face,
distinctive hair follicles, excessive eye discharge, swelling of eyelids,
visual disturbances, GI disturbances and jaundice. See M. Kuratsune et al., (EPA-560/6-75-004, 1976) p 14.
Potential symptoms of occupational overexposure are chloracne, dermal
lesions; hepatic injury; decreased pulmonary function; decreased birth
weight in offspring of exposed mothers; eye irritation (Safe, 1994).
See also Patty's Industrial Hygiene
and Toxicology vol. 2D, G. D. Clayton, F. E.
Clayton, Eds. (John Wiley & Sons, Inc., New York, 4th ed., 1994)
2433-2504. These substances are reasonably anticipated to be human
carcinogens: Report on Carcinogens, Eleventh
Edition (PB2005-104914, 2004) p III-218.Use
In electrical capacitors, electrical transformers,
gas-transmission turbines, vacuum pumps. Formerly used in U.S. as
hydraulic fluids, plasticizers, adhesives, fire retardants, wax extenders,
dedusting agents, pesticide extenders, inks, lubricants, cutting oils,
in heat transfer systems, carbonless reproducing paper.
