7741. Pridinol
Nomenclature
CAS number: 511-45-5
α,α-Diphenyl-1-piperidinepropanol; 1,1-diphenyl-3-piperidino-1-propanol; 3-piperidino-1,1-diphenyl-1-propanol; 1,1-diphenyl-3-(1-piperidyl)-1-propanol; 3-(N-piperidyl)-1,1-diphenyl-1-propanol; ridinol; C-238. C20H25NO; mol wt 295.42.
C 81.31%, H 8.53%, N 4.74%, O 5.42%.
Description and references
May be prepd from ethyl 1-piperidinepropionate and phenylmagnesium bromide: Adamson, GB 624118 (1949 to Wellcome Found.). Toxicity study: R. W. Cunningham et al., J. Pharmacol. Exp. Ther. 96, 151 (1949).
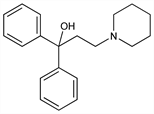
Properties
Crystals, mp 120-121°. Soluble in acetone.Derivative
Hydrochloride.Nomenclature
CAS number: 968-58-1
Parks 12 (Novartis). C20H25NO.HCl; mol wt 331.88.
C 72.38%, H 7.90%, N 4.22%, O 4.82%, Cl 10.68%.
Properties
Crystals, dec 238°. Sol in alc. LD50 in mice, rats (mg/kg): 35, 33 i.v.; 131, 91 i.p. (Cunningham).Derivative
Methanesulfonate.Nomenclature
CAS number: 6856-31-1
Pridinol mesylate; Konlax (Nippon Shinyaku); Loxeen (Tobishi); Lyseen (Novartis); Myoson (Strathmann). C20H25NO.CH3SO3H; mol wt 391.52.
C 64.42%, H 7.47%, N 3.58%, O 16.35%, S 8.19%.
Properties
Crystals, mp 152.5-155.0°. Sparingly sol in water.Therapeutic Category
Antiparkinsonian; muscle relaxant (skeletal).
Keywords
Pridinol Hydrochloride: Antiparkinsonian
Pridinol Mesylate: Muscle Relaxant (Skeletal)