7748. Primocarcin
Nomenclature
CAS number: 3750-26-3
5-(Acetylamino)-4-oxo-5-hexenamide; 4-acetamido-4-penten-3-one-1-carboxamide. C8H12N2O3; mol wt 184.19.
C 52.17%, H 6.57%, N 15.21%, O 26.06%.
Description and references
Antineoplastic antibiotic produced by the actinomycete, Nocardia fukaya. Isoln: Y. Sumiki et al., J. Antibiot. 13A, 416 (1960); Isono, Suzuki, ibid. 15A, 77 (1962). Isoln from Streptomyces diastatochromogens var. luteus: H. Abe et al., ibid. 20A, 167 (1967). Structure: Isono, ibid. 15A, 80 (1962). Synthesis: Bowman et al., J. Chem. Soc. 1965, 470.
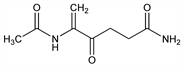
Properties
Needles from hot methanol, mp 130-131°. Sol in water, lower alcohols, acetone, pyridine; slightly sol in most other organic solvents. Aq soln stable from pH 5-8. uv max (water): 253 nm (ε 3420); in 0.1N NaOH: 215, 347 nm (ε 3230, 304). LD50 in mice (mg/kg): 50-60 i.p.; 300 i.v. (Isono, Suzuki).Derivative
Dihydroprimocarcin.C8H14N2O3; mol wt 186.21.
C 51.60%, H 7.58%, N 15.04%, O 25.78%.
Properties
Plates from acetone, mp 137-141°.Derivative
Tetrahydroprimocarcin.C8H16N2O3; mol wt 188.22.
C 51.05%, H 8.57%, N 14.88%, O 25.50%.