7786. Promethazine
Nomenclature
CAS number: 60-87-7
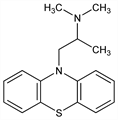
N,N,α-Trimethyl-10H-phenothiazine-10-ethanamine; 10-(2-dimethylaminopropyl)phenothiazine; 10-(2-dimethylamino-2-methylethyl)phenothiazine; N-(2′-dimethylamino-2′-methyl)ethylphenothiazine; proazamine; RP-3277; Fargan (Pfizer). C17H20N2S; mol wt 284.42.
C 71.79%, H 7.09%, N 9.85%, S 11.27%.
Description and references
Prepn from 10-phenothiazinepropyl chloride and dimethylamine in presence of Cu: Charpentier, Compt. Rend. 225, 306 (1947); US 2530451 (1950 to Rh^one-Poulenc); from Grignard complexes of dimethylaminopropyl halide and phenothiazine: Berg, Ashley, US 2607773 (1952 to Rh^one-Poulenc). Structure and isomerism: Edge, Wragg, J. Pharm. Pharmacol. 5, 279 (1953). Metabolic studies: Huang et al., J. Pharm. Sci. 59, 772 (1970). Toxicity: Rajsner, Collect. Czech. Chem. Commun. 34, 1019 (1969). Comprehensive description: C. M. Shearer, S. M. Miller, Anal. Profiles Drug Subs. 5, 429-465 (1976).
Properties
Crystals, mp 60°. bp3 190-192°.Derivative
Hydrochloride.Nomenclature
CAS number: 58-33-3
RP-3389; Atosil (Bayer); Farganesse (Pfizer); Fenazil (Sella); Hiberna (Mitsubishi); Lergigan (Recip); Phenergan (Sanofi-Aventis); Prothazin (Rodleben). C17H20N2S.HCl; mol wt 320.88.
C 63.63%, H 6.60%, N 8.73%, S 9.99%, Cl 11.05%.
Properties
Crystals from ethylene dichloride, mp 230-232° (some dec). Turns blue on prolonged exposure to air and moisture. uv max (water): 249, 297 nm (ε 28770, 3400). pH of 10% aq soln 5.3. Freely sol in water. Sol in alcohol, chloroform. Practically insol in acetone, ether, ethyl acetate. LD50 i.v. in mice: 55.0 mg/kg (Rajsner).Derivative
Compd with 8-chlorotheophylline.Nomenclature
CAS number: 17693-51-5
Promethazine teoclate; Avomine (Sanofi-Aventis). C17H20N2S.C7H7ClN4O2; mol wt 499.03.
C 57.76%, H 5.45%, N 16.84%, S 6.43%, Cl 7.10%, O 6.41%.
Therapeutic Category
Antihistaminic.
Therapeutic Category (Veterinary)
Antihistaminic, antiemetic, CNS depressant.Keywords
Antihistaminic; Tricyclics; Phenothiazines