7817. Propicillin
Nomenclature
CAS number: 551-27-9
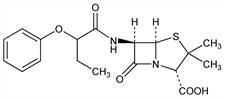
(2S,5R,6R)-3,3-Dimethyl-7-oxo-6-[(1-oxo-2-phenoxybutyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; 6-(α-phenoxybutyramido)penicillanic acid; α-phenoxypropylpenicillin; levopropylcillin. C18H22N2O5S; mol wt 378.44.
C 57.13%, H 5.86%, N 7.40%, O 21.14%, S 8.47%.
Description and references
Semi-synthetic antibiotic related to penicillin. Prepn from 6-aminopenicillanic acid: GB 877120 (1961 to Beecham); of salts: Perron et al., J. Am. Chem. Soc. 82, 3934 (1960); Glombitza, Ann. 673, 166 (1964). Metabolism in humans: M. Cole et al., Antimicrob. Agents Chemother. 3, 463 (1973). TLC determn: S. Hendrickx et al., J. Chromatogr. 291, 211 (1984).
Derivative
Potassium salt.Nomenclature
CAS number: 1245-44-9
BRL-284; PA-248; Baycillin (Bayer); Brocillin; Cetacillin; Oricillin (Grünenthal); Trescillin; Ultrapen. C18H21KN2O5S; mol wt 416.53.
C 51.90%, H 5.08%, K 9.39%, N 6.73%, O 19.21%, S 7.70%.
Properties
Crystals, dec 195-197°. Soluble at 20° in 1.2 parts water, 23 parts 95% (w/v) alcohol. pH of 1% w/v soln: 5-7.5.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins