Nomenclature
CAS number: 123-62-6
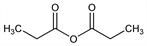
Propanoic acid 1,1′-anhydride; propanoic anhydride; propionyl oxide; methylacetic anhydride.
C
6H
10O
3; mol wt 130.14.
C 55.37%, H 7.75%, O 36.88%.
Description and references
Obtained by dehydration of the acid or by carbonylation
of its esters: Reppe, Friederich, US 2730546 (1956 to Badische Anilin- & Soda-Fabrik); from propionaldehyde by air oxidation in the presence of
cobalt and copper acetate catalysts: McFarlane, US 2491572 (1949 to Celanese). Other syntheses, e.g., from ethanol and carbon monoxide.
Toxicity study: H. F. Smyth et
al., Arch. Ind. Hyg. Occup. Med. 10, 61 (1954).
Properties
Liquid. Odor more pungent than that of the acid.
d40 1.0336; d415 1.0169; d420 1.0125; d440 0.98974; d450 0.97913. 8.4 lbs/gal at 20°. mp -45°. bp760 167.0°; bp400 146.0°; bp200 127.8°; bp100 107.2°; bp60 94.5°; bp40 85.6°; bp20 70.4°; bp10 57.7°; bp5 45.3°; bp1.0 20.6°. Flash pt, open cup: 165°F (74°C). nD17 1.4041; nD20 1.4038. Viscosity (cP): 1.144 at 20°;
0.978 at 30°; 0.853 at 40°; 0.7511 at 50°. Dec by water. Sol in
methanol, ethanol, ether, chloroform. LD50 orally in rats: 2.36 g/kg (Smyth).Use
Esterifying agent for certain perfume oils, fats,
oils, and especially cellulose. In the production of alkyd resins,
dyestuffs and drugs. Has been used as a dehydrating agent in some
sulfonations and nitrations.
