Nomenclature
CAS number: 57-55-6
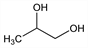
1,2-Propanediol; methyl glycol; 1,2-dihydroxypropane.
C
3H
8O
2; mol wt 76.09.
C 47.35%, H 10.60%, O 42.05%.
Description and references
Prepn from glycerol: Raschig, Prahl, Ber. 61, 185 (1928). Prepn of
levorotatory propylene glycol from hydroxyacetone by yeast reduction:
Levene, Walti, Org. Synth. coll. vol. II, 545 (1943). Prepn of S-(+)-form: E. Baer,
H. O. L. Fischer, J. Am. Chem. Soc. 70, 609 (1948); C. Melchiorre, Chem. Ind. (London) 1976, 218. Manuf from
propylene oxide by hydration: Faith, Keyes
& Clark's Industrial Chemicals, F. A. Lowenheim, M.
K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 688-691.
Toxicity data: W. Bartsch et al., Arzneim.-Forsch. 26, 1581 (1976). GC/MS determn
in plasma: C. Giachetti et al., Biomed. Environ. Mass Spectrom. 18, 592 (1989).
Review of toxicity, metabolism and biochemistry: Ruddick, Toxicol. Appl. Pharmacol. 21,
102 (1972); of toxicology and human exposure: Toxicological Profile for Ethylene Glycol and Propylene Glycol (US DHHS, PB98-101109, 1997) 250 pp.
Derivative
dl-Form.
Properties
Hygroscopic, viscous liquid. Slightly acrid taste.
mp -59°. bp760 188.2°; bp400 168.1°; bp200 149.7°; bp100 132.0°; bp60 119.9°; bp40 111.2°; bp20 96.4°; bp10 83.2°; bp5 70.8°; bp1.0 45.5°. d425 1.036. Flash pt, open cup: 210°F (99°C). nD20 1.4324. Miscible with water, acetone, chloroform. Sol in ether.
Will dissolve many essential oils, but is immiscible with fixed oils.
It is a good solvent for rosin. Under ordinary conditions propylene
glycol is stable, but at high temps it tends to oxidize giving rise
to products such as propionaldehyde, lactic acid, pyruvic acid and
acetic acid. LD50 orally in rats: 25 ml/kg (Bartsch).Derivative
l-Form.
Nomenclature
CAS number: 4254-14-2
(
R)-1,2-Propanediol.
Properties
bp12 88-90°, bp760 187-189°. [α]D20 -15.0°.Derivative
d-Form.
Nomenclature
CAS number: 4254-15-3
(
S)-1,2-Propanediol.
Properties
bp14 94-96°. bp765 186-188°. d25 1.04. nD23.5 1.4312. [α]D23 +20.1° (c = 7.5 in water). [α]D22 +4.2° (c = 6.6 in dry ethanol). [α]D20 +15.84° (neat).Use
Nontoxic antifreeze in breweries and dairy establishments.
Substitute for ethylene glycol and glycerol. In the manuf of synthetic
resins and de-icing solutions. Emulsifier in foods; solvent for food
colors and flavors. Pharmaceutic aid (humectant, solvent). As mist
to disinfect air; to create artificial smoke and mist for theatrical
use.
Therapeutic Category (Veterinary)
Glucogenic (orally) in ruminants.
