7882. Prosultiamine
Nomenclature
CAS number: 59-58-5
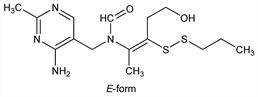
N-[(4-Amino-2-methyl-5-pyrimidinyl)methyl]-N-[4-hydroxy-1-methyl-2-(propyldithio)-1-butenyl]formamide; 2-(2-methyl-4-aminopyrimidin-5-yl)methylformamido-5-hydroxy-2-penten-3-yl
propyl disulfide; vitamin B1 propyl disulfide; thiamine propyl disulfide; dithiopropylthiamine; DTPT; TPD; Alinamin (Takeda); Binova (Gentili). C15H24N4O2S2; mol wt 356.51.
C 50.53%, H 6.79%, N 15.72%, O 8.98%, S 17.99%.
Description and references
Synthesis: Matsukawa, Kawasuki, J. Pharm. Soc. Jpn. 73, 216 (1953), C.A. 48, 2071 (1954); Matsukawa et al., J. Vitaminol. 1, 13 (1954); Fujiwara et al., US 2833768 (1958 to Takeda), cf. FR 1068459 (1954 to Takeda). Structural studies: Nishikawa et al., Chem. Pharm. Bull. 17, 932 (1969). Metabolism: Suzuoki-Ziro et al., J. Biochem. 58, 279 (1965); Nishikawa et al., J. Pharmacol. Exp. Ther. 157, 589 (1967).
Properties
Prisms from benzene, dec 128-129°. Sparingly soluble in water. Sol in organic solvents and lipids. Better absorbed upon oral ingestion by man, than thiamine hydrochloride.Derivative
Hydrochloride.Nomenclature
CAS number: 973-99-9
C15H24N4O2S2.HCl; mol wt 392.97.
C 45.85%, H 6.41%, N 14.26%, O 8.14%, S 16.32%, Cl 9.02%.
Properties
Crystals, dec 160-161°.Therapeutic Category
Vitamin (enzyme co-factor).
Keywords
Enzyme Cofactor; Vitamin/Vitamin Source; Vitamin B1