7899. Protoveratrines
Description and references
From the rhizome of Veratrum album L., Liliaceae: Salzberger, Arch. Pharm. 228, 462 (1890); Poethke, ibid. 275, 357 (1937); Craig, Jacobs, J. Biol. Chem. 143, 427 (1942); 149, 271 (1943). Mixture of protoveratrines A and B: Glen et al., Nature 170, 932 (1952); Klohs et al., J. Am. Chem. Soc. 74, 5107 (1952); Nash, Brooker, ibid. 75, 1942 (1953); Stoll, Seebeck, Helv. Chim. Acta 36, 718 (1953); Nash, Brooker, US 2929812 (1960 to Allied Labs). Structure of protoveratrines A and B: Kupchan, Ayres, J. Am. Chem. Soc. 82, 2252 (1960). Comparative toxicity: O. Krayer et al., J. Pharmacol. Exp. Ther. 82, 167 (1944); K. Tanaka, ibid. 113, 89 (1955).
Properties
Slightly bitter, sternutative crystals from alc, dec 266-267°. [α]D25 -38.6° (pyridine). [α]D25 -8.5° (c = 1.99 in chloroform). Soluble in chloroform; dil aq acidic solns; slightly sol in ether. Practically insol in water, petr ether. LD50 i.v. in mice: 0.048 mg/kg (Krayer).Derivative
Protoveratrine A.Nomenclature
CAS number: 143-57-7
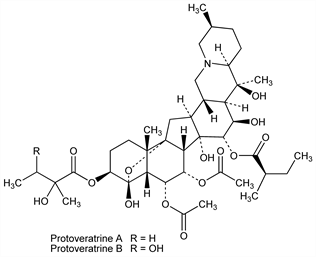
[3β(S),4α,6α,7α,15α(R),16β]-4,9-Epoxycevane-3,4,6,7,14,15,16,20-octol
6,7-diacetate 3-(2-hydroxy-2-methylbutanoate) 15-(2-methylbutanoate); Protalba. C41H63NO14; mol wt 793.94.
C 62.02%, H 8.00%, N 1.76%, O 28.21%.
Properties
Crystals from acetone, dec 267-269°. [α]D25 -40.5° (pyridine); [α]D25 -10.5° (chloroform). Soluble in chloroform, pyridine, hot alcohol. LD50 s.c. in male mice: 0.29 mg/kg (Tanaka).Derivative
Protoveratrine B.Nomenclature
CAS number: 124-97-0
[3β(2R,3R),4α,6α,7α,15α(R),16β]-4,9-Epoxycevane-3,4,6,7,14,15,16,20-octol
6,7-diacetate 3-(2,3-dihydroxy-2-methylbutanoate) 15-(2-methylbutanoate); veratetrine; neoprotoveratrine. C41H63NO15; mol wt 809.94.
C 60.80%, H 7.84%, N 1.73%, O 29.63%.
Properties
Crystals from acetone, dec 268-270°. [α]D25 -37° (pyridine); [α]D25 -3.5° (chloroform). Soluble in chloroform, pyridine, hot alcohol. LD50 s.c. in male mice: 0.21 mg/kg (Tanaka).Derivative
Mixture of protoveratrine A and B.Nomenclature
Provell (Lilly); Tensatrin (Gedeon Richter).Therapeutic Category
Antihypertensive.
Keywords
Antihypertensive