7912. Pseudococaine
Nomenclature
Description and references
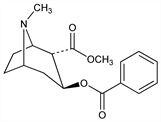
Cocaine diastereomer with greater local anesthetic activity than the natural substance. Synthesis: Einhorn, Marquardt, Ber. 23, 468, 981 (1890); Willst"atter, Bode, Ann. 326, 42 (1903); Willst"atter, Bommer, ibid. 422, 34 (1921); Willst"atter et al., ibid. 434, 138 (1923); GB 210050 (1923 to E. Merck). Configuration: S. P. Findlay, J. Am. Chem. Soc. 76, 2855 (1954). Conformational analysis and 1H, 13C NMR studies: F. I. Carroll et al., J. Org. Chem. 1982, 13. Pharmacokinetics: A. L. Misra et al., Experientia 32, 895 (1976). Interaction with sodium channels: J C. Matthews, A. Collins, Biochem. Pharmacol. 32, 455 (1983). Pharmacology of cocaine and pseudococaine: G. Schmidt et al., Arch. Exp. Pathol. Pharmakol. 240, 523 (1961).
Properties
Prisms, mp 47°. [α]D20 +42° (c = 5 in chloroform). Freely sol in ether, chloroform, benzene, petr ether. Slightly sol in water.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from alcohol, mp 210°. [α]D20 +41° (c = 5), less sol in water than cocaine hydrochloride.Derivative
Tartrate.Nomenclature
Properties
Crystals, mp 139°. [α]D20 +43° (c = 5 in water). Sol in 4 parts water, in alcohol. The aq soln is stable and may be sterilized by boiling, without decompn. pH about 3.7 (2% soln).Derivative
Sodium tartrate.Nomenclature
Psicaine N.Properties
More sol in water than the tartrate; esp useful where a neutral soln is desired.Derivative
n-Propyl ester analog.Nomenclature
Therapeutic Category
Formerly as anesthetic (local).
Keywords
Anesthetic (Local)