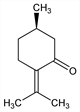
7937. Pulegone
Nomenclature
Description and references
Found in oils derived from plants of the Labiatae family as (+)-form. Readily isolated in quantity from the pennyroyal oils from Mentha pulegium L., M. longifolia (L.) Huds., and Hedeoma pulegioides (L.) Pers., Labiatae: Gildemeister, Hoffmann, Die "atherischen Ole Vol. I, p 560 (1928); Simonsen, The Terpenes Vol. I (2nd ed, 1947) p 370. Synthesis: Kuhn, Schinz, Helv. Chim. Acta 36, 161 (1953). Synthesis of the (±)-form: Black et al., J. Chem. Soc. 1956, 2971; Wolinsky et al., J. Org. Chem. 30, 3207 (1965); of the S(-)-form: E. J. Corey et al., ibid. 41, 380 (1976). Improved synthesis of the R(+)-form: T. Sato et al., Tetrahedron Lett. 1980, 3377. Conversion of the R(+) to the S(-)-form: H. E. Ensley, R. V. C. Carr, ibid. 1977, 513. Biosynthetic study: A. Akhila, D. V. Banthorpe, Z. Pflanzenphysiol. 99, 277 (1980), C.A. 94, 2073r (1981).