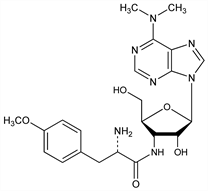
7943. Puromycin
Nomenclature
Description and references
Aminonucleoside antibiotic produced by the soil actinomycete Streptomyces alboniger. Structural analog of aminoacyl-tRNA; inhibits protein synthesis. Isoln: J. N. Porter et al., Antibiot. Chemother. 2, 409 (1952); eidem, US 2763642 (1956 to Am. Cyanamid). Structure: C. W. Waller et al., J. Am. Chem. Soc. 75, 2025 (1953); P. W. Fryth et al., ibid. 80, 2736 (1958). Synthesis: B. R. Baker et al., ibid. 76, 4044 (1954); 77, 12 (1955); M. J. Robins et al., J. Org. Chem. 66, 8204 (2001). Conformation: O. Jardetzky, J. Am. Chem. Soc. 85, 1823 (1963). Effect on protein synthesis: D. Nathans, Proc. Natl. Acad. Sci. USA 51, 585 (1964). Use in gene transfer studies: S. de la Luna et al., Gene 62, 121 (1988). Review: D. Nathans in Antibiotics vol. I, D. Gottlieb, P. D. Shaw, Eds. (Springer-Verlag, New York, 1967) pp 259-277.