8000. Pyrogallol
Nomenclature
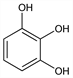
CAS number: 87-66-1
1,2,3-Benzenetriol; 1,2,3-trihydroxybenzene; pyrogallic acid. C6H6O3; mol wt 126.11.
C 57.14%, H 4.80%, O 38.06%.
Description and references
Observed by Scheele in 1786; prepd by Braconot in 1818. Prepn from gallic acid: Marsh, GB 144897 (1919); Rinderknecht, Niemann, J. Am. Chem. Soc. 70, 2605 (1948); from p-tert-butylphenol: Stevens, US 2603662 (1952 to Gulf). Synthesis from aliphatic sources: Shipchandler et al., J. Chem. Soc. Perkin Trans. 1 1975, 1400. Isoln from Penicillium patulum: Tanenbaum, Bassett, Biochim. Biophys. Acta 28, 21 (1958). Acute toxicity: J. W. Dollahite et al., Am. J. Vet. Res. 23, 1264 (1962).
Properties
White, odorless crystals, mp 131-133°. Becomes grayish on exposure to air and light. Poisonous. d 1.45; bp 309°. Sublimes when slowly heated. One gram dissolves in 1.7 ml water, 1.3 ml alc, 1.6 ml ether; slightly sol in benzene, chloroform, carbon disulfide. The aq soln darkens on exposure to air, quite rapidly when alkaline. Keep well closed and protected from light. Incompat. Alkalies, ammonium hydroxide, antipyrine, camphor, phenol, menthol. LD50 orally in rabbits: 1.6 g/kg (Dollahite).Derivative
Monoacetate.Nomenclature
CAS number: 1330-51-4
Eugallol (Knoll). C8H8O4; mol wt 168.15.
C 57.14%, H 4.80%, O 38.06%.
Properties
White or brownish liquid, bp23 ≈185°. Sol in water, alcohol, chloroform, ether, acetone and castor oil. Marketed as a 67% soln in acetone.Derivative
Triacetate.Nomenclature
CAS number: 525-52-0
Acetpyrogall; Lenigallol (Knoll). C12H12O6; mol wt 252.22.
C 57.14%, H 4.80%, O 38.06%.