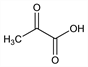
8021. Pyruvic Acid
Nomenclature
Description and references
Intermediate in sugar metabolism and in enzymatic carbohydrate degradation (alcoholic fermentation) where it is converted to acetaldehyde and CO2 by carboxylase, see Nord, Chem. Rev. 26, 423 (1940). In muscle, pyruvic acid (derived from glycogen) is reduced to lactic acid during exertion, which is reoxidized and partially retransformed to glycogen during rest. The liver can convert pyruvic acid to alanine by amination. Pyruvic acid has been isolated from cane sugar fermentation broth by fixation with β-naphthylamine giving α-methyl-β-naphthocinchonic acid: Grab, Biochem. Z. 123, 84 (1921). The most practical method of preparing pyruvic acid is by distillation of tartaric acid in presence of potassium acid sulfate as dehydrating agent. The distillate must be fractionated under reduced pressure: Erlenmeyer, Ber. 14, 321 (1881); D"obner, Ann. 242, 269 (1887); Howard, Fraser, Org. Synth. coll. vol. I (2nd ed., 1941) p 475. Absorption spectrum: Henri, Fromageot, Bull. Soc. Chim. [4] 37, 846 (1925).