8028. Quatrimycin
Nomenclature
CAS number: 79-85-6
[4R-(4α,4aβ,5aβ,6α,12aβ)]-4-(Dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenecarboxamide; epitetracycline. C22H24N2O8; mol wt 444.43.
C 59.45%, H 5.44%, N 6.30%, O 28.80%.
Description and references
Prepn by epimerization of tetracycline: McCormick et al., J. Am. Chem. Soc. 79, 2849 (1957); Kaplan et al., Antibiot. Chemother. 7, 569 (1957); Remmers et al., J. Pharm. Sci. 52, 752 (1963). Acute toxicity: L. Kung, H. Sun, Yao Hsueh Hsueh Pao 13, 244 (1966), C.A. 65, 7869e (1966).
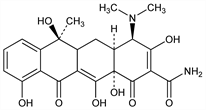
Derivative
Monohydrate.Properties
Crystals, mp 178° dec. [α]D25 -335° (c = 0.5 in 0.03N HCl). uv max (0.01N H2SO4): 216, 255, 270, 355 (ε 13,900, 16,400, 15,200, 14,700). LD50 i.v. in mice: 85.8 mg/kg (Kung, Sun).Derivative
Ammonium salt monohydrate.C22H27N3O8.H2O; mol wt 479.48.
C 55.11%, H 6.10%, N 8.76%, O 30.03%.
Properties
Yellow crystals, mp 170° dec. [α]D25 -321° (c = 0.5 in 0.03N HCl). Soly in water >50 mg/kg.Derivative
Hydrochloride.C22H25ClN2O8; mol wt 480.90.
C 54.95%, H 5.24%, Cl 7.37%, N 5.83%, O 26.62%.
Properties
Amorphous yellow solid. [α]D25 -325° (c = 0.58 in 0.2N HCl). Soly in water >100 mg/ml.Derivative
Methiodide.C23H27IN2O8; mol wt 586.37.
C 47.11%, H 4.64%, I 21.64%, N 4.78%, O 21.83%.