8064. Quinizarin
Nomenclature
CAS number: 81-64-1
1,4-Dihydroxy-9,10-anthracenedione; 1,4-dihydroxyanthraquinone; C.I. 58050. C14H8O4; mol wt 240.21.
C 70.00%, H 3.36%, O 26.64%.
Description and references
Prepn from p-chlorophenol and phthalic anhydride: Reynolds, Bigelow, J. Am. Chem. Soc. 48, 420 (1926); US 1845632, C.A. 26, 2203; Org. Synth. coll. vol. I, 476 (New York, 1941). Prepn from hydroquinone: Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 299. Also prepd from diazotized p-chloroaniline and phthalic anhydride: GB 373999, C.A. 27, 3946 (1933); by treating anthraquinone with ammonium persulfate in sulfuric acid: Wacker, J. Prakt. Chem. [2] 54, 90 (1896). Purification from contaminating purpurin: Org. Synth. (loc. cit.). See also: Colour Index vol. 4 (3rd ed., 1971) p 4515.
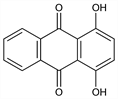
Properties
Orange crystals from acetic acid, mp 200-203° (Org. Syn.). Orange plates from ether. Deep red needles from alcohol, benzene, toluene, xylene. mp 196°. Sublimes in high vacuum. Absorption spectrum: Meek, Watson, J. Chem. Soc. 109, 544 (1916); Meek, ibid. 111, 969 (1917). pK (18°) 9.51. Moderately sol in alcohol with red color. Sol in ether with brown color and yellow fluorescence. Sol with violet color in aq alkalies and in ammonia. Black precipitate with CO2. One gram dissolves in about 13 g of boiling glacial acetic acid.Derivative
Dimethyl ether.C16H12O4; mol wt 268.26.
C 71.64%, H 4.51%, O 23.86%.