8116. Rasagiline
Nomenclature
Description and references
Irreversible MAO-B inhibitor. Prepn: M. B. H. Youdim et al., EP 436492; eidem. US 5457133 (1991, 1995 both to Teva). Pharmacology: J. P. M. Finberg, M. B. H. Youdim, Neuropharmacology 43, 1110 (2002); of metabolites: S. Glezer, J. P. M. Finberg, Eur. J. Pharmacol. 472, 173 (2003). Mechanism of action: M. B. H. Youdim et al., Biochem. Pharmacol. 66, 1635 (2003). Double-blind clinical trial as adjunctive therapy to levodopa: J. M. Rabey et al., Clin. Neuropharmacol. 23, 324 (2000); multicenter trial in early Parkinson's disease: Arch. Neurol. 59, 1937 (2002); in levodopa-treated Parkinson's patients: Parkinson Study Group, ibid. 62, 241 (2005); O. Rascol et al., Lancet 365, 947 (2005). Review of pharmacology: J. J. Chen, D. M. Swope, J. Clin. Pharmacol. 45, 878-894 (2005); of clinical development: O. Rascol, Expert Opin. Pharmacother. 6, 2061-2075 (2005); F. Stocchi, Int. J. Clin. Pract. 60, 215-221 (2006).
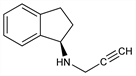
Derivative
Methanesulfonate.Nomenclature
Therapeutic Category
Antiparkinsonian.
Keywords
Antiparkinsonian