8131. Remifentanil
Nomenclature
CAS number: 132875-61-7
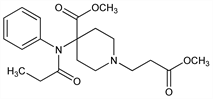
4-(Methoxycarbonyl)-4-[(1-oxopropyl)phenylamino]-1-piperidinepropanoic
acid methyl ester; 4-carboxy-4-(N-phenylpropionamido)-1-piperidinepropionic
acid dimethyl ester; remifentanyl; GI-87084. C20H28N2O5; mol wt 376.45.
C 63.81%, H 7.50%, N 7.44%, O 21.25%.
Description and references
Synthetic mu-opioid agonist; derivative of fentanyl, q.v. Prepn: P. L. Feldman et al., EP 383579; eidem, US 5019583 (1990, 1991 both to Glaxo); eidem et al., J. Med. Chem. 34, 2202 (1991). Opioid receptor activity: M. K. James et al., J. Pharmacol. Exp. Ther. 259, 712 (1991). LC-MS determn in plasma: E. Bossu et al., J. Pharm. Biomed. Anal. 42, 367 (2006). Review of pharmacokinetics and therapeutic use: R. Beers, E. Camporesi, CNS Drugs 18, 1085-1104 (2004); of pharmacology and clinical use in general anesthesia: L. J. Scott, C. M. Perry, Drugs 65, 1793-1823 (2005); in analgesia-based sedation: A. J. Battershill, G. M. Keating, ibid. 66, 365-385 (2006).
Properties
Oil. Sol in methanol, ethyl acetate. pKa 7.26.Derivative
Hydrochloride.Nomenclature
CAS number: 132539-07-2
GI-87084B; Ultiva (Glaxo Wellcome). C20H28N2O5.HCl; mol wt 412.91.
C 58.18%, H 7.08%, N 6.78%, O 19.37%, Cl 8.59%.
Properties
Crystals from methanol + ether, mp 212-214°.Note
This is a controlled substance (opiate): 21 CFR, 1308.12.Therapeutic Category
Analgesic; anesthesia adjunct.
Keywords
Analgesic; Opioids; Phenylpiperidines