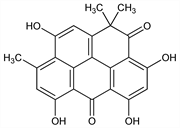
8152. Resistomycin
Nomenclature
CAS number: 20004-62-0
3,5,7,10-Tetrahydroxy-1,1,9-trimethyl-2H-benzo[cd]pyrene-2,6(1H)-dione; X-340. C22H16O6; mol wt 376.36.
C 70.21%, H 4.29%, O 25.51%.
Description and references
Unusually stable antibiotic substance produced by Streptomyces resistomycificus: Brockmann, Schmidt-Kastner, Naturwissenschaften 38, 479 (1951); Ber. 87, 1460 (1954); Brockmann, DE 888918 (1953). Structure: Brockmann, Angew. Chem. 76, 863 (1964). Revised structure: Bailey et al., Chem. Commun. 1968, 374; Brockmann et al., Ber. 102, 1224 (1969). Synthetic studies: J. F. Kingston, L. Weiler, Can. J. Chem. 55, 785 (1977); K. James, R. A. Raphael, Tetrahedron Lett. 1979, 3895. Total synthesis: B. A. Keay, R. Rodrigo, J. Am. Chem. Soc. 104, 4725 (1982). Isoln from Streptomyces griseoflavus B71, 13C-NMR and biosynthesis: G. H"ofle, H. Wolf, Ann. 1983, 835.