8158. Resveratrol
Nomenclature
Description and references
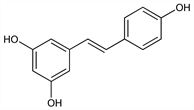
Phytoalexin found in a variety of plants; active ingredient of Asian folk medicine “Kojo-Kon” which is the powdered root of the Japanese knotweed. Isoln from white hellebore and structure: M. Takaoka, J. Fac. Sci., Hokkaido Imp. Univ. Ser. 3 3, 1 (1940); C.A. 34, 7887 (1940); from fescue grass: R. G. Powell et al., Phytochemistry 35, 335 (1994). Isoln of monomers, oligomers, isomers and glucosides from wine: F. Mattivi et al., J. Agric. Food Chem. 43, 1820 (1995). GC-MS determn: D. M. Goldberg et al., J. Am. Enol. Viticult. 46, 159 (1995). Produced as stress metabolite in response to fungal infection or injury: G. H. Dai et al., Physiol. Mol. Plant Pathol. 46, 177 (1995); P. Jeandet et al., J. Phytopathol. 143, 135 (1995). Antiplatelet aggregating activity: A. A. E. Bertelli et al., Int. J. Tissue React. 17, 1 (1995); and implications in coronary heart disease: C. R. Pace-Asciak et al., Clin. Chim. Acta 235, 207 (1995).
Properties
Off-white powder from MeOH, mp 253-255°.Derivative
3-β-mono-d-glucoside.Nomenclature
Description and references
Isoln from fresh root of Polygonum cuspidatum Sieb. & Zucc., Polygonaceae, and structure: Nonomura et al., Yakugaku Zasshi 83, 988 (1963).
Derivative
(Z)-Form.Nomenclature
Description and references
GC-MS determn in wine: D. M. Goldberg et al., J. Agric. Food Chem. 43, 1245 (1995).