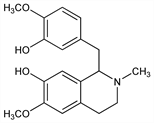
8163. Reticuline
Nomenclature
Description and references
Precursor of many aporphine and morphine-type alkaloids. Isoln of d-form from Anona reticulata Linn., Anonaceae: Gopinath et al., Ber. 92, 776 (1959); dl-form from opium: Brochmann-Hanssen, Furuya, J. Pharm. Sci. 53, 575 (1964); Planta Med. 12, 328 (1964). Structure: Brochmann-Hanssen, Nielson, Tetrahedron Lett. 1965, 1271. Configuration: Battersby, Evans, ibid. 1275. Synthesis of amorphous dl-form: Gopinath et al., Ber. 92, 1657 (1959); P. Kerekes et al., Acta Chim. Acad. Sci. Hung. 98, 491 (1978). Synthesis of crystalline dl-form: Baxter et al., J. Chem. Soc. C 1965, 3645; Chan, Maitland, ibid. 1966, 753; K. C. Rice, A. Brossi, J. Org. Chem. 45, 592 (1980); G. Dornyei et al., Tetrahedron Lett. 1982, 2913. Asymmetric synthesis of (S)-reticuline: Konda et al., Chem. Pharm. Bull. 23, 1063 (1975). Biosynthesis: D. S. Dewan et al., J. Chem. Soc. Perkin Trans. 1 1977, 1662; A. Barrett et al., ibid. 1979, 652.