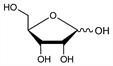
8204. d-Ribose
Nomenclature
Description and references
Prepd by hydrolysis of yeast-nucleic acid: Levene, Jacobs, Ber. 42, 1201, 3247 (1909); Levene, Clark, J. Biol. Chem. 46, 19 (1921); Bredereck, Ber. 71, 408 (1938); Bredereck et al., ibid. 73, 956 (1940); Phelps, US 2152662 (1939 to U.S. Gov't); by ion-exchange resin chromatography: Cohn, Science 109, 377 (1949); J. Am. Chem. Soc. 71, 2275 (1949); 72, 1471 (1950). From glucose: Karrer, Helv. Chim. Acta 18, 1435 (1935); Austin, Humoller, J. Am. Chem. Soc. 56, 1152 (1934); Kuhn et al., Ber. 68, 1765 (1935); from nucleosides: Laufer, Charney, US 2379913; US 2379914 (both 1945 to Schwarz Labs.); from d-erythrose: Sowden, J. Am. Chem. Soc. 72, 808 (1950); from l-glutamic acid: Koga et al., Tetrahedron Lett. 1971, 263. Reduction of d-ribonic acid: van Ekenstein, Blanksma, Chem. Zentralbl. 1913, II, 1562; Steiger, Helv. Chim. Acta 19, 189 (1936). Review: Overend, Stacey, in Nucleic Acids vol. I, E. Chargaff, J. N. Davidson, Eds. (Academic Press, New York, 1955) pp 1-80.