8290. Ruboxistaurin
Nomenclature
Description and references
Protein kinase Cβ (PKCβ) inhibitor; macrocyclic bisindolylmaleimide. Prepn: W. F. Heath, Jr. et al., EP 657458; eidem, US 5552396 (1995, 1996 both to Eli Lilly); M. R. Jirousek et al., J. Med. Chem. 39, 2664 (1996); M. M. Faul et al., J. Org. Chem. 63, 1961 (1998). Characterization of salt forms: G. L. Engel et al., Int. J. Pharm. 198, 239 (2000). Enantioselective synthesis: B. M. Trost, W. Tang, Org. Lett. 3, 3409 (2001). Antiangiogenic activity: R. P. Danis et al., Invest. Ophthalmol. Visual Sci. 39, 171 (1998). Clinical trial in diabetic retinopathy: PKC-DRS Study Group, Diabetes 54, 2188 (2005); in diabetic neuropathy: A. I. Vinik et al., Clin. Ther. 27, 1164 (2005); in diabetic nephropathy: K. R. Tuttle et al., Diabetes Care 28, 2686 (2005). Review of pharmacology and therapeutic potential: S. V. Joy et al., Ann. Pharmacother. 39, 1693-1699 (2005); A. Vinik, Expert Opin. Invest. Drugs 14, 1547-1559 (2005).
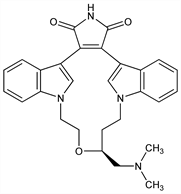
Properties
uv max (ethanol): 493, 382, 282, 232, 204 nm (ε 3944, 3796, 9946, 38382, 43887). Soly in water: <1 μg/ml.Derivative
Hydrochloride.Nomenclature
Properties
Orange cake. [α]D25 -28.7° (c = 1.0 in ethanol). Soly in water: 0.1 mg/ml.Derivative
Methanesulfonate monohydrate.Nomenclature
Properties
Thin red or red-orange plates from acetone/water. Dec 270°. Soly in water: 0.5 mg/ml.Therapeutic Category
In treatment of diabetic microvascular complications.