8294. Rufloxacin
Nomenclature
Description and references
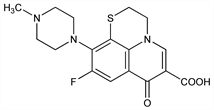
Fluorinated quinolone; structurally similar to ofloxacin, q.v. Prepn: G. Mascellani et al., AU 85 39142; eidem, US 4684647 (1985, 1987 both to Mediolanum); V. Cecchetti et al., J. Med. Chem. 30, 465 (1987). In vitro activity: R. Wise et al., Antimicrob. Agents Chemother. 29, 649 (1992). Mechanism of action and resistance: L. J. V. Piddock et al., J. Antimicrob. Chemother. 31, 855 (1993). Pharmacokinetics and safety: J. C. Kisicki et al., ibid. 36, 1296 (1992). HPLC determn in biological fluids: F. Lombardi et al., J. Chromatogr. 576, 129 (1992). Clinical study in cystitis and urinary tract infections: R. Mattina et al., Infection 21, 106 (1993); in bronchitis: W. Klietmann et al., Antimicrob. Agents Chemother. 37, 2298 (1993).
Derivative
Hydrochloride.Nomenclature
Properties
Crystals from ethanol/water, mp 322-324° (Cecchetti). LD50 in rats, mice (mg/kg): 285, 224 i.v.; LD50 in rabbits, male, female rats (mg/kg): 660, 631, 501 orally (Cecchetti).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Quinolones and Analogs