8340. Salsoline
Nomenclature
CAS number: 101467-40-7
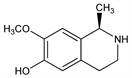
(R)-1,2,3,4-Tetrahydro-7-methoxy-1-methyl-6-isoquinolinol; 6-hydroxy-7-methoxy-1-methyl-1,2,3,4-tetrahydroisoquinoline; (+)-salsoline. C11H15NO2; mol wt 193.24.
C 68.37%, H 7.82%, N 7.25%, O 16.56%.
Description and references
In Salsola richteri Karel., Chenopodiaceae. Extraction procedure: A. Orechoff, N. Proskurnina, Ber. 66, 841 (1933); N. Proskurnina, A. Orekhoff, Bull. Soc. Chim. Fr. 1937, 1265. Structure and synthesis: E. Sp"ath et al., Ber. 67, 1214 (1934); O. Kovács, G. Fodor, Ber. 84, 795 (1951). Abs config: A. R. Battersby, T. P. Edwards, J. Chem. Soc. 1960, 1214. Biosynthetic studies: I. McFarlane, M. Slaytor, Phytochemistry 11, 235 (1972). Synthesis of enantiomers: S. Teitel et al., J. Med. Chem. 17, 134 (1974). HPLC study: J. Str"ombom, J. G. Bruhn, J. Chromatogr. 147, 513 (1978).
Properties
Crystals from alc, mp 221°. [α]D20 +34.5° (c = 1 in 0.1N HCl). Sol in chloroform, hot alc, dil NaOH. Slightly sol in water, benzene. Almost insol in ether, petr ether.Derivative
Hydrochloride.Nomenclature
CAS number: 51424-33-0
Properties
mp 174-175°. [α]D +31.0° (methanol). uv max (isopropanol): 204, 227, 284, 286 nm (ε 39400, 5900, 3540, 3530). Sol in water, hot alcohol. Very sparingly sol in acetone, chloroform. LD50 in mice (mg/kg): 140 i.v.; >1000 orally (Teitel).Derivative
(-)-Form.Nomenclature
CAS number: 89-31-6
(S)-1,2,3,4-Tetrahydro-7-methoxy-1-methyl-6-isoquinolinol.