8373. Sarcosine
Nomenclature
CAS number: 107-97-1
N-Methylglycine; N-methylaminoacetic acid; Sar; N-methylglycocoll; methylaminoethanoic acid. C3H7NO2; mol wt 89.09.
C 40.44%, H 7.92%, N 15.72%, O 35.92%.
Description and references
Has been found in starfish and sea urchins: A. Kossel, S. Edlbacher, Z. Physiol. Chem. 94, 264 (1915); in rock lobsters: L. Novellie, H. M. Schwartz, Nature 173, 450 (1954). Formation from caffeine by dec with barium hydroxide: Paulmann, Arch. Pharm. 232, 601 (1894). Prepd on a large scale from formaldehyde, sodium cyanide, and methylamine: Eschweiler, Ann. 279, 39 (1894); Baumann, J. Biol. Chem. 21, 563 (1915); Caverly, US 2720540 (1955 to du Pont); Leake, Brakebill, US 3009954 (1961 to Allied Chem.). Synthesis: M. Ebata et al., Bull. Chem. Soc. Jpn. 39, 2535 (1966). Review of syntheses: T. Shirai, Synthetic Production and Utilization of Amino Acids, T. Kaneko et al., Eds. (Wiley, New York, 1974) pp 184-186.
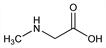
Properties
Orthorhombic, deliquescent crystals from dil methanol. Dec 212°. Sweetish taste. pK1′ 2.23; pK2′ 10.01. Sol in water: 5 ml of a satd aq soln contain 2.1412 g sarcosine. Slightly sol in alc.Derivative
Sodium salt.Nomenclature
CAS number: 4316-73-8
C3H6NNaO2; mol wt 111.08.
C 32.44%, H 5.44%, N 12.61%, Na 20.70%, O 28.81%.
Properties
Usually sold as aq soln, pH 12.0.Derivative
Hydrochloride.Nomenclature
CAS number: 637-96-7
C3H7NO2.HCl; mol wt 125.55.
C 28.70%, H 6.42%, N 11.16%, O 25.49%, Cl 28.24%.